Open Access | Research article
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
CASC15 Promotes Lens Epithelial Cell Apoptosis in Age-Related Cataracts by Regulating MiR-139-5p/caspase-3
#These authors contributed equally
*Corresponding author: Xiu-Hua Guo
Mailing address: Department of Ophthalmology, Miyun Teaching
Hospital Affiliated to Capital Medical University. 383 Yangguang
Street, Miyun District, Beijing, 101500, China.
E-mail: 1006055546@qq.com
Received: 25 March 2019 Accepted: 28 May 2019
DOI: 10.31491/CSRC.2019.06.032
Abstract
Objective: The aim of this study was to reveal the role of CASC15 and miR-139-5p function in regulating agerelated cataracts (ARC).
Methods: The expression of CASC15, miR-139-5p and caspase-3 were detected by qRT-PCR. Western blot was used to analyze the level of apoptosis-related proteins. Flow cytometry was used to measure the apoptosis of SAR01/04l cells treated with UV irradiation.
Results: The expression of CASC15 and caspase-3 in the lens of ARC was higher than that in normal lens, but the expression of miR-139-5p had opposite trend. The level of caspase-3 and Bax were increased and Bcl-2 was decreased in ARC group. In addition, CASC15 negatively regulated miR-139-5p expression and promoted SAR01/04l cell apoptosis. CASC15 could combine with miR-139-5p, and miR-139-5p could combine with caspase-3 3’-UTR. Furthermore, pcDNA-caspase-3 could reverse the inhibitory effect of si-CASC15 and miR- 139-5p mimic on SRA01/04 cell apoptosis.
Conclusion: CASC15 promoted lens epithelial cell apoptosis by regulating miR-139-5p/caspase-3.
Keywords
ARC; CASC15; LECs apoptosis; miR-139-5p
Introduction
Cataract is a common eye disease, which is the leading
cause of visual impairment and blindness [1,2]. Age-related cataract (ARC) is the most common types of cataracts,
which often occurs in people aged 50 years and over, and
is characterized by loss of lens transparency [3]. Recently, with the aging of the population intensified, the incidence of ARC has increased, and most of the new cases
come from low-income countries [4]. ARC is a complex
multifactorial disease caused by the interaction of genes
and environmental factors [5]. Several studies found that
risk factors such as age, sex, smoking, diabetes, sunlight
or ultraviolet B radiation, vitamins and body mass index
(BMI) were related to the occurrence and development
of ARC [6]. In addition, genetic background also plays an
important role in the development of ARC [7]. At present, the surgical treatment of ARC has achieved good
results worldwide, but the etiology of ARC is not clearly understood. A large number of studies have shown
that the apoptosis of lens epithelial cells (LECs) is the
common cytological basis for the formation of cataracts
except congenital cataract [8]. Therefore, to investigate
the mechanism of LECs apoptosis is important for the
prevention and treatment of ARC.
Long non-coding RNA (lncRNA) is a non-coding RNA
with a length greater than 200 nucleotides [9]. Studies
have shown that lncRNAs play an important role in Dosage compensation effect, epigenetic regulation, cell cycle
regulation and cell differentiation regulation [10]. Moreover, lncRNAs are closely related to the development of
tumor [11]. Cancer susceptibility candidate 15 (CASC15)
is a lncRNA and expresses in brain [12]. Recently, studies
have suggested that CASC15 played an important role in
neuroblastoma [12] and melanoma [13], etc, but the role of
CASC15 in LECs apoptosis has not been reported.
MicroRNAs (miRNAs) are endogenously expressed
non-coding RNAs, which are widely involved in cell proliferation, apoptosis and autophagy [14]. Recent studies
have shown that miRNAs play an important role in the
regulation of LECs apoptosis [15]. Chen et al. [16] found that
miR-34a could promote LECs apoptosis through E2F3
pathway. Zhang et al.[17] suggested that downregulation
of miR-133b could inhibit LECs apoptosis. MiR-139-5p
is a relatively widespread miRNA in human cells. It has
been found that miR-139-5p can be used as a biomarker for cancer [18]. Chen et al. [19] found that miR-139-5p
inhibited cell proliferation of uterine fibroids. Song et al.[20] reported that miR-139-5p inhibited the metastasis
of colorectal cancer. However, the effects of miR-139-5p
on the apoptosis of LECs and its regulation mechanism
have not been reported. LncBase Predicted v.2 software
predicted that CASC15 combined with miR-139-5p, and
TargetScan software predicted that miR-139-5p could
combine with 3’-UTR of caspase-3. Caspase-3 is an important factor for cell apoptosis. Previous studies have
demonstrated that caspase-3 plays a key role in LECs
apoptosis [21,22]. Thus, we speculated that the role of
CASC15 and miR-139-5p on LECs apoptosis might be
related with caspase-3.
In this study, we first observed the expression of CASC15,
miR-139-5p and apoptosis-related proteins in the lens
tissues of patients with ARC and SRA01/04 cells, and
then investigated the regulation mechanism of CASC15/
miR-139-5p/caspase-3 on LECs apoptosis. The aim of
this study was to reveal the relationship between CASC15
and ARC, and to provide a new target for the prevention
and treatment of blinding eye disease.
Materials and Methods
Samples
In this study, we selected 25 ARC patients who had been excluded other eye diseases. It included 15 males and 10 females, and with the average age of 60.7±5.3 years (aged from 50 to 70). In all cases, continuous curvilinear capsulorhexis was used to obtain the anterior capsule of the lens in cataract surgery. Then, eye anterior lens capsules transparent specimens of 25 adults without cataract were collected as the control group. It included 14 males and 11 females, and with the average age of 59.2±5.4 years (aged from 50 to 70). These lenses from postmortem eyes were obtained with 8 to 24 hours after death. These specimens were provided by the Miyun Teaching Hospital Affiliated to Capital Medical University. The study protocol was approved by the Ethics Committee of the Miyun Teaching Hospital Affiliated to Capital Medical University, and all patients were informed consent.
Cell culture
Human LECs line - SRA01/04 cells (purchased from the cell bank of Chinese Academy of Medical Sciences) were cultured in DMEM medium (Gibco, USA) supplemented with 10% FBS (Gibco, USA), 100 U/ml of penicillin and 100 μg/ml of streptomycin, and incubated at 37°C in an incubator with 5% CO2.
Ultraviolet (UV) irradiation
Apoptosis model of HLEC was established by ultraviolet irradiation. The cells were irradiated with an ultraviolet (UV) lamp (XX-15B, Spectroline, Westbury, USA) [23]. The irradiation intensity was 360 μW/cm2 , and the irradiation time was 30min.
Cell transfection
Si-CASC15, si-caspase-3, si-NC, pcDNA-CASC15, pcDNA, miR-139-5p inhibitor and inhibitor-NC were respectively transfected into SRA01/04 cells using Lipofectamine 2000 (Invitrogen, USA) following the manufacturer’s instructions.
Quantitative real-time PCR (qRT-PCR)
According to the manufacturer’s instructions, the total
RNA was extracted with Trizol Kit (Invitrogen, USA), and
then reverse transcribed into cDNA for qRT-PCR. The
expression of CASC15, miR-139-5p and caspase-3 were
measured using the 2-ΔΔCt method.
The primer sequences:
MiR-139-5p F: 5’-TCTACAGTGCACGTGTCTCCAGT-3’
MiR-139-5p R: 5’-TGGAGACACGTGCACTGTAGATT-3’
Caspase-3 F: 5’-TAC CTC AAC TTC GAC ATC AGG-3’
Caspase-3 R: 5’-CAG TGG AAA AGC TGT ACT GC-3’
CASC15 F: 5’-CGCCGGGGTATCTCCCTCTCG-3’
CASC15 R: 5’-CATTTCCCCCGCTGCAGTCCA-3’.
Western blot
Total protein was extracted using the protein lysis buffer (Solarbio, Beijing). Then, the protein samples were resolved by 10% SDS-PAGE and transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Bedford, USA). The membranes were incubated with the primary antibodies against caspase-3 (1: 500, Abcam, Chicago, IL, USA), Bax (1: 1000, Abcam), Bcl-2 (1: 1000, Abcam) and β-actin (1:1000, Abcam) overnight at 4°C, and then incubated with secondary antibody peroxidase conjugated goat rabbit anti-IgG (Solarbio, Beijing). Quantity one software was used to quantify the band intensity.
TUNEL assay
Terminal dexynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay was used to measure cell apoptosis in tissue samples [24]. Paraffin-embedded eye anterior lens capsules transparent specimens were cut into tissue sections of 5 µm, and fixed in 4% (w/v) paraformaldehyde. Before the TUNEL assay, the samples were permeated with 0.1% (v/v) TritonX-100 (Beyotime, Shanghai, China), and blocked with 3% (v/v) H2O2. The TUNEL reaction mixture was then dropped onto the surface of the samples. After incubating at 37°C in a dark and humid environment for one hour, converter-POD was dropped onto the surface of the samples, and incubated for 30 min at 37°C followed by diaminobenzidine substrate. The samples were counterstained with hematoxylin, analyzed under a fluorescence microscope. The percentage of TUNEL-positive cells was used to calculate the apoptosis rate.
Flow cytometry analysis
Cell apoptosis was detected using flow cytometry analysis. According to the manufacturer’s instructions, Annexin V-EGFP/PI (apoptosis detection kit, Research Science, Shanghai) was used to measure cells apoptosis. The samples were analyzed by flow cytometry (Becton Dickinson, CA). Then, the data were analyzed using Cell Quest software.
Luciferase reporter assay
The miR-139-5p binding site of caspase-3 was amplified by PCR. Then, caspase-3 3’UTR fragment or mutant caspase-3 3’UTR fragment were cloned into vector pcDNA with a firefly luciferase reporter gene (Beyotime, China). The miR-139-5p mimic or inhibitor or negative controls (NC) were respectively transfected into SRA01/04 cells. After 48h transfection, the cells were collected for measurement of luciferase activity.
RNA-binding protein immunoprecipitation (RIP) assay
According to the manufacturer’s instructions, RIP assay was performed using RIP Kit (Sigma-Aldrich, Shanghai). The cells were lysed in RIP buffer and incubated with anti-Ago2 antibody (Sigma-Aldrich, Shanghai). IgG was used as a negative control. Trizol reagent (Invitrogen, USA) was used to extract precipitated RNA. The level of CASC15 and miR-139-5p was analyzed by qRT-PCR.
RNA pull-down assay
RNA pull-down assay was performed by Magnetic RNA-Protein Pull-Down Kit (Thermo Fisher Scientific, USA) following the manufacturer’s instructions. Western blot was used to detect the level of Ago2, and qRTPCR was used to identify the level of miR-139-5p.
Statistical analysis
The experiments were repeated at least three times and analyzed by using SPSS 19.0 software. The data were presented as mean ± standard deviation (SD). P<0.05 was considered as statistical significance.
Results
The expression of CASC15, miR-139-5p and apoptosis-related protein in the crystalline lens of ARC
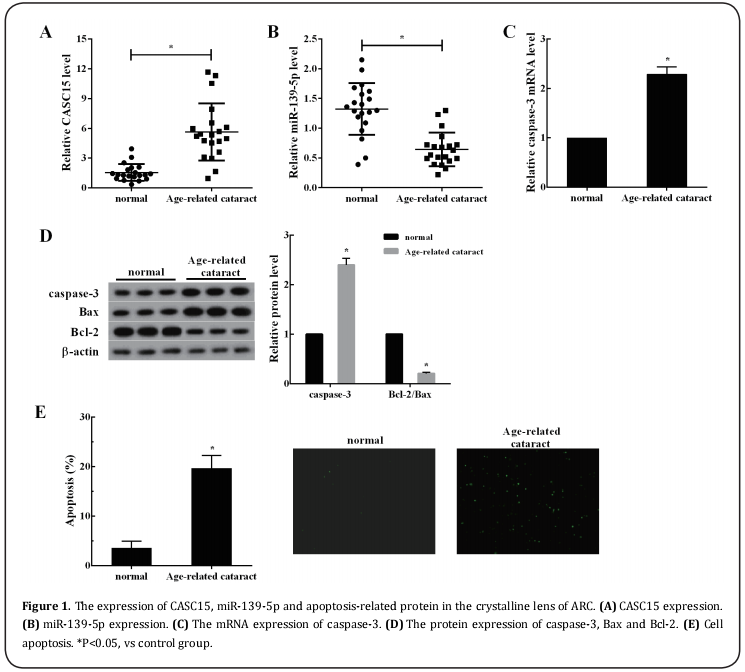
qRT-PCR results showed that the CASC15 expression in the crystalline lens of ARC was markedly higher than that in the normal crystalline lens (Figure 1A), but the expression of miR-139-5p had an opposite trend (Figure 1B). Moreover, the mRNA expression of caspase-3 was increased significantly in the crystalline lens of the ARC compared with the normal crystalline lens (Figure 1C). The protein expression of caspase-3 and Bax were increased and Bcl-2 expression was decreased markedly in the ARC group compared with the normal group (Figure 1D). In addition, the cell apoptotic rate in ARC group was higher than that in normal group (Figure 1E).
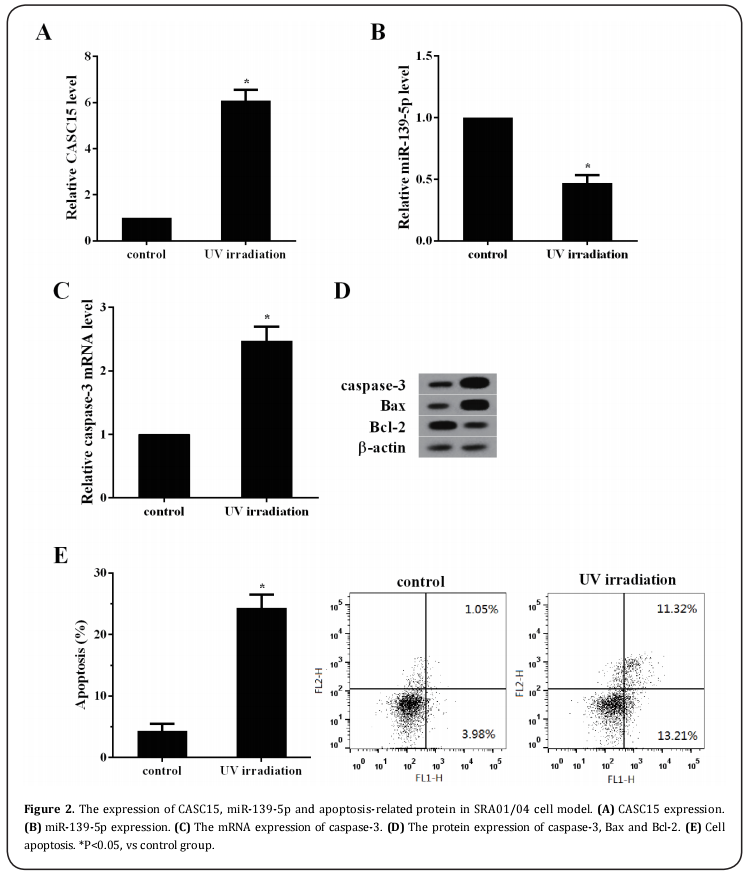
The expression of CASC15, miR-139-5p and apoptosis-related protein in SRA01/04 cell model
As shown in Figure 2A&B, compared with the control cells, the expression of CASC15 in SRA01/04 cells treated with UV irradiation was markedly increased and the miR-139-5p expression was significantly decreased. Moreover, the mRNA expression of caspase-3 and the protein expression of caspase-3 and Bax were significantly increased in UV group, and Bcl-2 expression was significantly decreased compared with control group (Figure 2C&D). In addition, as shown in Figure 2E, UV irradiation markedly increased SRA01/04 cell apoptotic rate.
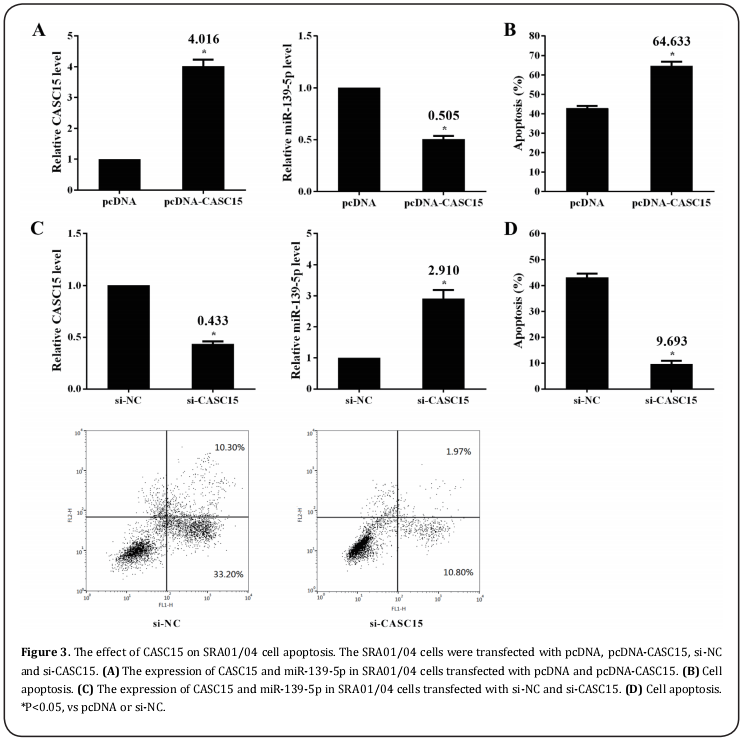
LnCASC15 promoted SRA01/04 cell apoptosis
The SRA01/04 cells were transfected with pcDNA, pcDNA-CASC15, si-NC and si-CASC15 respectively, and were treated with UV irradiation for 30mins. The results showed that pcDNA-CASC15 could promote the expression of CASC15 and inhibit the expression of miR-139-5p (Figure 3A). On the other hand, overexpression of CASC15 significantly promoted the apoptosis of SRA01/04 cells (Figure 3A&B). However, si-CASC15 reversed the trend. si-CASC15 could decrease CASC15 expression and increase miR-139-5p expression, and inhibit cell apoptosis (Figure 3C&D).
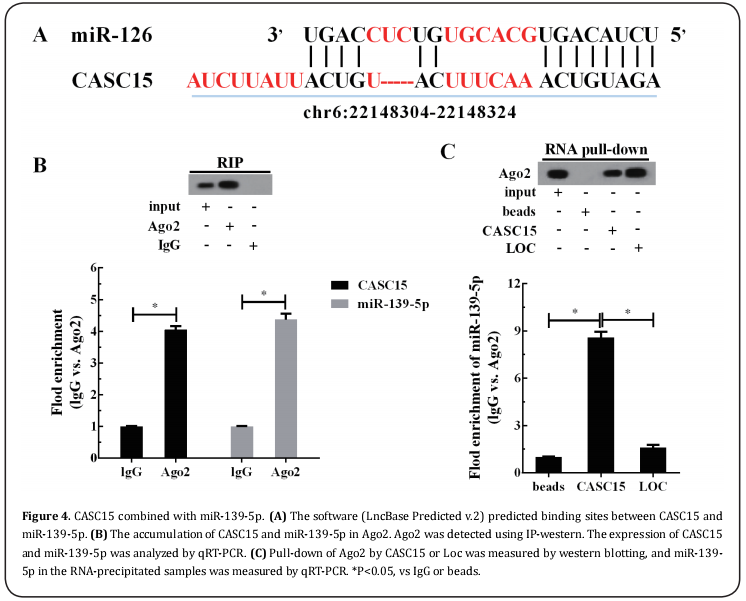
CASC15 combined with miR-139-5p
The software (LncBase Predicted v.2) predicted that CASC15 combined with miR-139-5p (Figure 4A). As shown in Figure 4B, Ago2 antibody precipitated the Ago2 protein from the cell lysates, and compared with IgG control, higher CASC15 and miR-139-5p in the Ago2 pellet was detected by qRT-PCR. Moreover, RNA pulldown assay showed that CASC15 interacted with Ago2 directly (Figure 4C). Compared with control, the content of miR-139-5p in CASC15 pulled down pellet was significantly increased, and the content of miR-139-5p in Loc (the positive control of CASC15) pulled down pellet was only slightly increased (Figure 4C).
CASC15 negatively regulated miR-139-5p expression in SRA01/04 cells
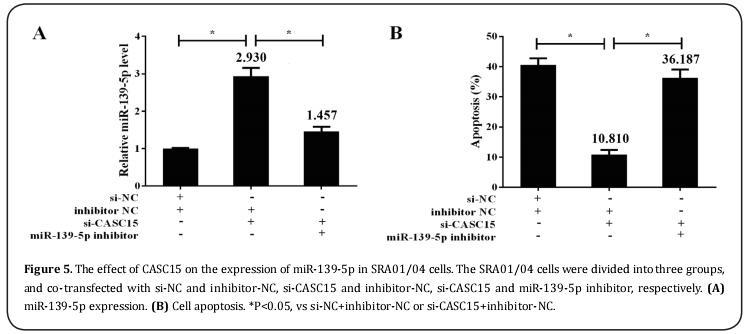
The SRA01/04 cells were divided into three groups, and co-transfected with si-NC and inhibitor-NC, si-CASC15 and inhibitor-NC, si-CASC15 and miR-139-5p inhibitor, respectively. The results showed that si-CASC15 up-regulated miR-miR-139-5p expression (Figure 5A) and inhibited SRA01/04 cell apoptosis (Figure 5B). However, miR-139-5p inhibitor could reverse the trend.
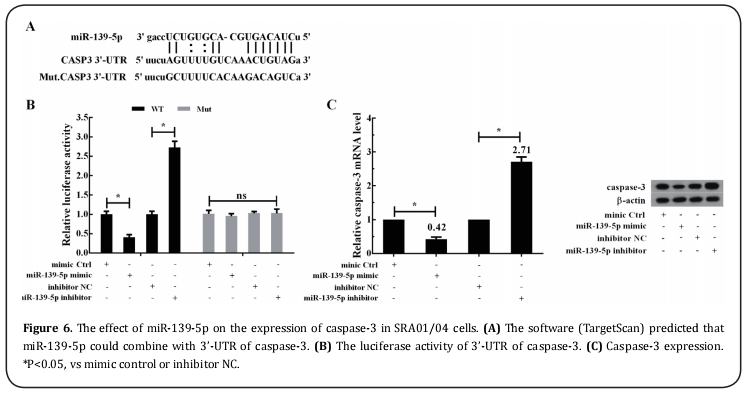
The effect of miR-139-5p on the expression of caspase-3
The software (TargetScan) predicted that miR-139-5p could combine with 3’-UTR of caspase-3 (Figure 6A). As shown in Figure 6B, in caspase-3’URT-WT group, miR-139-5p mimic could reduce the luciferase activity compared with mimic control, and miR-139-5p inhibitor could markedly enhance the luciferase activity compared with inhibitor control. However, the luciferase activity had no significant change in caspase-3’URT-MUT group. In addition, as shown in Figure 6C, SRA01/04 cells were divided into four groups: miR-139-5p mimic group, mimic control group, miR-139-5p inhibitor group, and inhibitor control group. The results showed that the mRNA and protein expression of caspase-3 in the miR- 139-5p mimic group was decreased compared with the mimic control group, while the caspase-3 expression in the miR-139-5p inhibitor group was increased compared with the inhibitor control group.
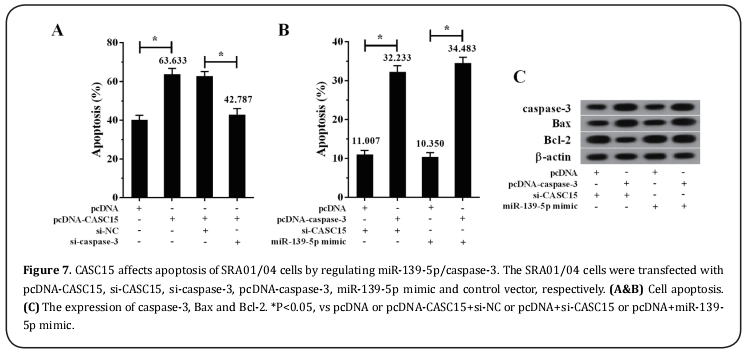
CASC15 promoted apoptosis of SRA01/04 cells by regulating miR-139-5p/caspase-3
As shown Figure 7A, SRA01/04 cells were transfected with si-caspase-3, pcDNA-CASC15 and their controls. The results showed that the SRA01/04 cell apoptotic rate was increased in the pcDNA-CASC15 group compared with the pcDNA group, and decreased in the pcDNA-CASC15+si-caspase-3 group compared with the pcDNA-CASC15+si-NC group. As shown Figure 7B, SRA01/04 cells were transfected with pcDNA-caspase-3, miR-139-5p mimic, si-CASC15 and their controls. The results showed that the SRA01/04 cell apoptotic rate was increased in the pcDNA-caspase-3+si-CASC15 group compared with the pcDNA+si-CASC15 group, and the cell apoptotic rate was also increased in the pcDNAcaspase-3+miR-139-5p mimic group compared with the pcDNA+miR-139-5p mimic group. Moreover, compared with pcDNA+si-CASC15 and pcDNA+miR-139-5p mimic, the expression of caspase-3 and Bax in SRA01/04 cells transfected with pcDNA-caspase-3 was increased, but the Bcl-2 expression was decreased.
Discussion
ARC, known as senile cataract, is the most common reason of blindness in elder people. Although the surgery is
still the most effective treatment for cataract [25], the huge
medical expenditure of surgical treatment also brings
heavy burden to health resources and social economy
of our country. Thus, it is imperative to explore new approaches to non-surgical treatment of cataract.
In this study, we showed that CASC15 and caspase-3
were up-regulated in lens tissues of ARC and UV irradiation-induced SAR01/04 cells, but miR-139-5p was
down-regulated. Moreover, we found that overexpression CASC15 could decrease miR-139-5p expression and
promote apoptosis of SAR01/04l cells. These results indicated that CASC15, miR-139-5p and caspase-3 might be associated with LECs apoptosis in ARC. Therefore, we
further studied the mechanism of CASC15, miR-139-5p
and caspase-3 in ARC.
LncRNAs are associated with various eye diseases. Xu et
al. [26] found that Vax2os1 and Vax2os2 were specifically
expressed in intra-ocular retinal neovascularization. Yan
et al. [27] reported that down-regulation of lncRNA-MIAT
could reduce the formation of retinal neovascularization induced by diabetes. Young et al. [28] suggested that
knockdown of TUG1 could inhibit the migration of developing rod cells in the outer nuclear layer. The present
study found that CASC15 played an important role in
ARC. Moreover, CASC15 could be combined miR-139-5p,
and knockdown of CASC15 could increase miR-139-5p
expression and inhibit SAR01/04l cell apoptosis. These
results suggested CASC15 played a role in LECs apoptosis by regulating miR-139-5p expression.
In recent years, a lot of data reported that miRNAs played
an important role in the development and progression
of cataract. Hughes et al. [29] found that the mutation of
miR-184 seed region could lead to anterior polar cataract. Qin et al. [30] reported that miR-125b inhibited LECs
apoptosis in patients with ARC by targeting silencing
p53. In addition, other studies have shown that miR-
31, miR-99a/b, and let-7b are differentially expressed
in lens tissues of ARC [31]. In this study, we also found
that miR-139-5p was abnormally expressed in lens of
patients with cataract. Furthermore, we found that miR-
139-5p could directly target the 3’UTR of caspase-3 and
negatively regulate caspase-3 expression. Caspase-3 is a
key protease involved in mammalian cell apoptosis [32].
Recently, studies found that the levels of caspase-3 and
Bax in the lens of patients with cataract were increased
and the Bcl-2 level was decreased [33], which was consistent with our results. Hence, we speculated that miR-
139-5p might exert its function in ARC by regulating the
expression of caspase-3. Further studies confirmed this
hypothesis. In SRA01/04 cells treated with UV irradiation, overexpression caspase-3 could reverse the inhibitory effect of knockdown of CASC15 and miR-139-5p
mimic on SRA01/04 cell apoptosis.
In conclusion, our data indicated that CASC15 could
promote LECs apoptosis by regulating miR-139-5p/
caspase-3, which provided a new strategy for the treatment of ARC.
References
1. Zheng, S. J., Orsini, N., Ejdervik, L. B., and Wolk, A. (2014)
Long-term physical activity and risk of age-related
cataract: a population-based prospective study of male
and female cohorts. Ophthalmology 122, 274
2. Petrash, J. M. (2013) Aging and age-related diseases
of the ocular lens and vitreous body. Investigative
Ophthalmology & Visual Science 54, 54-59
3. Song, E., Sun, H., Xu, Y., Ma, Y., Zhu, H., and Pan, C. W. (2014)
Age-Related Cataract, Cataract Surgery and Subsequent
Mortality: A Systematic Review and Meta-Analysis. Plos
One 9, e112054
4. Rao, G. N., Khanna, R., and Payal, A. (2011) The global
burden of cataract. Current Opinion in Ophthalmology
22, 4-9
5. West, S. (2007) Epidemiology of Cataract:
Accomplishments over 25 years and Future Directions.
Ophthalmic Epidemiol 14, 173-178
6. Wang, A., Han, J., Jiang, Y., and Zhang, D. (2014) Association
of vitamin A and β-carotene with risk for age-related
cataract: A meta-analysis. Nutrition 30, 1113-1121
7. Liu, X. C., Liu, X. F., Hu, Z. D., and Li, Z. H. (2015)
Polymorphisms of DNA repair genes XPD (Lys751Gln) and
XRCC1 (Arg399Gln), and the risk of age-related cataract: a meta-analysis. Current Eye Research 40, 676-682
8. Li, W. C., Kuszak, J. R., Dunn, K., Wang, R. R., Ma, W., Wang, G.
M., Spector, A., Leib, M., Cotliar, A. M., and Weiss, M. (1995)
Lens epithelial cell apoptosis appears to be a common
cellular basis for non-congenital cataract development in
humans and animals. Journal of Cell Biology 130, 169-181
9. Spizzo, R., Almeida, M. I., Colombatti, A., and Calin, G. A.
(2012) Long non-coding RNAs and cancer: a new frontier
of translational research? Oncogene 31, 4577-4587
10. Fatica, A., and Bozzoni, I. (2014) Long non-coding RNAs:
new players in cell differentiation and development.
Nature Reviews Genetics 15, 7-21
11. Gibb, E. A., Brown, C. J., and Wan, L. L. (2011) The functional
role of long non-coding RNA in human carcinomas.
Molecular Cancer 10, 38
12. Russell, M. R., Penikis, A., Oldridge, D. A., Alvarezdominguez,
J. R., Mcdaniel, L., Diamond, M., Padovan, O., Raman, P., Li,
Y., and Wei, J. S. (2015) CASC15-S Is a Tumor Suppressor
lncRNA at the 6p22 Neuroblastoma Susceptibility Locus.
Cancer Research 75, 3155-3166
13. Lessard, L., Liu, M., Marzese, D. M., Wang, H., Chong, K.,
Kawas, N., Donovan, N. C., Kiyohara, E., Hsu, S., and Nelson,
N. (2015) The CASC15 long intergenic non-coding RNA
locus is involved in melanoma progression and phenotypeswitching. Journal of Investigative Dermatology 135,
2464-2474
14. Abba, M., Patil, N., and Allgayer, H. (2014) MicroRNAs in
the Regulation of MMPs and Metastasis. Cancers 6, 645
15. Klettner, A. (2015) Age-related macular degeneration -
biology and treatment. Medizinische Monatsschrift Für
Pharmazeuten 38, 258-264; quiz 265-256
16. Chen, W. (2015) MiR-34a induce human lens epithelial
cell apoptosis through E2F3 pathway. Investigative
Ophthalmology & Visual Science 56, 2640
17. Zhang, F., Meng, W., and Tong, B. (2016) Down-Regulation of
MicroRNA-133b Suppresses Apoptosis of Lens Epithelial
Cell by Up-Regulating BCL2L2 in Age-Related Cataracts.
Medical Science Monitor International Medical Journal of
Experimental & Clinical Research 22, 4139-4145
18. Zhang, H. D., Jiang, L. H., Sun, D. W., Li, J., and Tang, J. H.
(2015) MiR-139-5p: promising biomarker for cancer.
Tumor Biology 36, 1355-1365
19. Chen, H., Xu, H., Meng, Y. G., Zhang, Y., Chen, J. Y., and Wei, X.
N. (2016) miR-139-5p regulates proliferation, apoptosis,
and cell cycle of uterine leiomyoma cells by targeting
TPD52. Oncotargets & Therapy 9, 6151-6160
20. Song, M., Yin, Y., Zhang, J., Zhang, B., Bian, Z., Quan, C.,
Zhou, L., Hu, Y., Wang, Q., and Ni, S. (2014) MiR-139-5p
inhibits migration and invasion of colorectal cancer by
downregulating AMFR and NOTCH1. Protein & Cell 5,
851-861
21. Yao, H., Tang, X., Shao, X., Feng, L., Wu, N., and Yao, K.
(2007) Parthenolide protects human lens epithelial cells
from oxidative stress-induced apoptosis via inhibition of
activation of caspase-3 and caspase-9. Cell research 17,
565-571
22. Yao, K., Wang, K., Xu, W., Sun, Z., Shentu, X., and Qiu, P.
(2003) Caspase-3 and its inhibitor Ac-DEVD-CHO in rat
lens epithelial cell apoptosis induced by hydrogen in vitro.
Chinese medical journal 116, 1034-1038
23. Osada, H., Yoshitake, Y., Ikeda, T., Ishigaki, Y., Takata,
T., Tomosugi, N., Sasaki, H., and Yonekura, H. (2011)
Ultraviolet B-induced expression of amphiregulin and
growth differentiation factor 15 in human lens epithelial
cells. Molecular Vision 17, 159-169
24. Jiang, Y., Fu, R., Zhao, J., Wu, D., Qiao, G., Li, R., and Zhang,
J. (2015) Effects of ELL-associated factor 2 on ultraviolet
radiation-induced cataract formation in mice. Molecular
medicine reports 12, 6605-6611
25. Rosen, E. S. (2014) Age-related macular degeneration and
cataract surgery. Journal of Cataract & Refractive Surgery
40, 173-174
26. Xu, X. D., Li, K. R., Li, X. M., Yao, J., Qin, J., and Yan, B.
(2014) Long non-coding RNAs: new players in ocular
neovascularization. Molecular Biology Reports 41, 4493-
4505
27. Yan, B., Yao, J., Liu, J. Y., Li, X. M., Wang, X. Q., Li, Y. J., Tao, Z.
F., Song, Y. C., Chen, Q., and Jiang, Q. (2015) lncRNA-MIAT
regulates microvascular dysfunction by functioning as a
competing endogenous RNA. Circulation Research 116,
1143-1156
28. Young, Tracy, L., Cepko, and Constance, L. (2004) A Role
for Ligand-Gated Ion Channels in Rod Photoreceptor
Development. Neuron 41, 867-879
29. Hughes, A. E., Bradley, D. T., Campbell, M., Lechner, J.,
Dash, D. P., Simpson, D. A., and Willoughby, C. E. (2011)
Mutation altering the miR-184 seed region causes familial
keratoconus with cataract. American Journal of Human
Genetics 89, 628-633
30. Qin, Y., Zhao, J., Min, X., Wang, M., Luo, W., Wu, D., Yan,
Q., Li, J., Wu, X., and Zhang, J. (2014) MicroRNA-125b
inhibits lens epithelial cell apoptosis by targeting p53 in
age-related cataract. Biochimica Et Biophysica Acta 1842,
2439-2447
31. Peng, C. H., Liu, J. H., Woung, L. C., Lin, T. J., Chiou, S. H.,
Tseng, P. C., Du, W. Y., Cheng, C. K., Hu, C. C., and Chien, K. H.
(2012) MicroRNAs and cataracts: correlation among let-7
expression, age and the severity of lens opacity. British
Journal of Ophthalmology 96, 747
32. Odonkor, C. A., and Achilefu, S. (2009) Modulation of
Effector Caspase Cleavage Determines Response of Breast
and Lung Tumor Cell Lines to Chemotherapy. Cancer
Investigation 27, 417-429
33. Ji, Y., Cai, L., Zheng, T., Ye, H., Rong, X., Rao, J., and Lu, Y. (2015)
The mechanism of UVB irradiation induced-apoptosis in
cataract. Molecular and Cellular Biochemistry 401, 87-95