Open Access | Case Report
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Acute massive gastric dilatation: a surgical emergency
*Corresponding author: Dr. Parthasarathi Hota
Mailing address: Department of General Surgery, Calcuta Natonal Medical College, Gorachand Road, Kolkata – 700014, India.
Email: psh1011@rediffmail.com
Received: 10 September 2019 Accepted: 25 October 2019
DOI: 10.31491/CSRC.2019.12.043
Abstract
A 40 year old patient presents with acute pain abdomen with abdominal distension. History of unusual heavy meal one day before and following which symptoms appear. Few episodes of vomiting were associated. Resuscitation done. Straight x ray shows dilated gastric shadow. Patient posted for laparotomy after failing of conservative measures. On opening abdomen, hugely distended stomach seen with thinned out gastric wall and patchy areas of discolouration. A side to side gastrojejunostomy done after decompression on the dependent stomach. Post-operative recovery was uneventful. Psychological evaluation did not reveal any abnormality. Acute gastric dilatation can cause mucosal necrosis and gastric perforation therefore early diagnosis and gastric decompression is the key. Though most patients respond to conservative measures, failing which surgical decompression is needed.
Keywords
Acute gastric dilatation; eating disorders; gastric decompression; gastrojejunostomy; acute massive gastric dilatation
Introduction
Acute gastric dilatation [AGD] is a rare disorder with
most of the references in the literature as case reports.
AGD is encountered most often as a postoperative complication in abdominal surgery and in a multitude of
disorders, such as anorexia and bulimia nervosa, psychogenic polyphagia, trauma, diabetes mellitus etc. [1-5].
Acute massive gastric dilatation [AMGD] is the extreme
form of AGD. In literature the demarcation of AGD and
AMGD is not clearly mentioned. When the stomach is
extremely distended occupying the abdomen from
diaphragm to pelvis and from left to right, the AGD is
referred to as AMGD. Most frequently AMGD requires
surgical intervention to prevent or to treat gastric necrosis [3].
We present a case of acute massive gastric dilatation
with thinned out gastric wall but without perforation.
After failure of conservative measures, the patient was
treated surgically.
Case Report
A 40 year old male patient came to the emergency department at night with complaints of acute abdominal
pain and distension, multiple episodes of vomiting and
obstipation for 2 days.
The patient was apparently well until 2 days ago when
after having an unaccustomed heavy lunch he started
complaining of abdominal distension which was not
relieved by any means but aggravated with food and
fluid intake. The distension progressed gradually with
abdominal pain and multiple episodes of vomiting. The
vomitus comprised of food materials, non- bilious, nonprojectile and vomiting did not relieved the distension.
There was no history of hematemesis or fever. There
was history of obstipation for which he received enema
at the primary health centre, referred thereafter to our
hospital.
The patient had no history of alcohol/tobacco addiction.
He had normal bowel and bladder habits with one episode of binge eating two days ago. There was no history
of any chronic illness like diabetes or hypertension. He
was not taking any medication and did not undergo any
surgery before.
On general examination, the patient had no signs of anaemia, oedema, jaundice, clubbing or cyanosis. He was of
average built and nutritional status. At the time of admission his pulse was 110 per minute and blood pressure
was 110/70 mmHg.
On local examination, the abdomen was hugely distended with no dilated veins, scar mark and the umbilicus
everted. There was generalised tenderness. There was
no ascites or any abdominal lump. There was moderate amount of guarding and rigidity but no rebound tenderness.
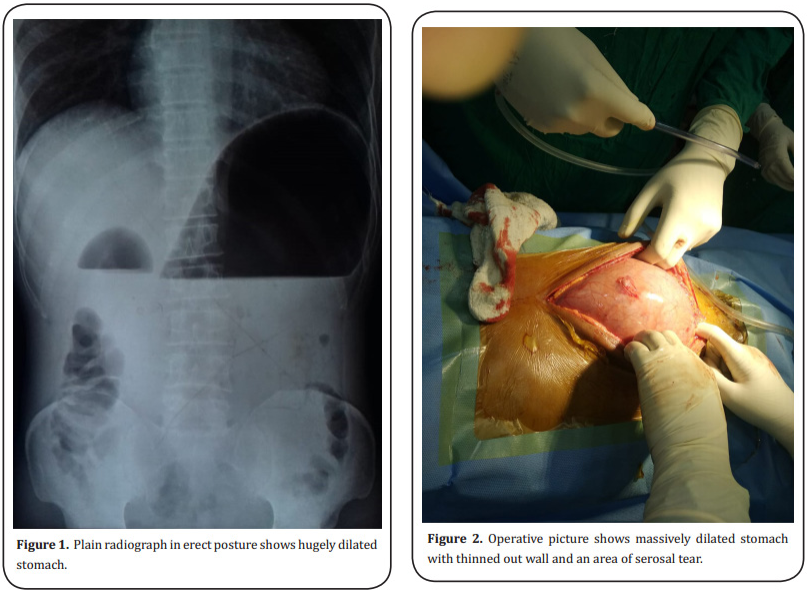
After admission the patient was resuscitated with normal saline and urgent upright abdominal x-ray was
done. The x-ray shows grossly dilated stomach (Figure
1) but there was no evidence of free gas under the diaphragm. A large bore nasogastric tube was inserted
and careful suctioning done. Scanty food materials and
fluid came out but then the material stopped coming
out of the stomach. The nasogastric tube repositioned
and changed but still without any yield. Then the NG tube was subjected to continuous vacuum suction pump
but still nothing evacuated. The abdominal distension
did not improve and the patient’s condition gradually
worsened. The abdominal pain was not relieved either.
At this stage conservative measures abandoned and the
patient was posted for emergency laparotomy after taking high risk consent. Standard midline vertical incision
given. As soon as the peritoneum opened, hugely distended stomach was seen extending from xiphisternum
to below umbilicus going towards the pelvis. The stomach wall was thinned out but there was no perforation
or obvious necrosis but a few patchy areas of slightly
dusky discolouration seen with one area of serosal tear (Figure 2). There was no rotation of the stomach and the
rest of the bowel was normal. Liver, spleen was normal,
there was no fluid inside the peritoneal cavity and no
other lump noted.
Posterior, retrocolic, isoperistaltic side to side gastro-jejunostomy done in two layers incorporating the gastrotomy. Primary repair done for the serosal tear with absorbable suture.
Full psychological evaluation of the patient done in the post-operative period but no abnormality including any eating disorders was found.
The recovery was uneventful with the patient passing flatus on third day and discharged on satisfactory condition after 7 days. Upper GI endoscopy was performed after 6 weeks and it shows normal gastric mucosa and with good and healthy anastomosis and no other abnormal finding.
Discussion
In 1833, Duplay first described acute gastric dilatation
[1]. Acute ischemic necrosis of stomach is a very rare disease due to its abundant vascular supply. In experimental
animals, in order to produce ischemic necrosis, closure
of the right and left gastric and gastroepiploic arteries
together with at least 80% blockage of the collaterals
is required [2]. The important causes are postoperative
complications [3, 4], anorexia nervosa and bulimia, psychogenic polyphagia, diabetes mellitus, trauma, electrolyte
disturbances, gastric volvulus, and spinal conditions [1,
5–10].
Our patient was of average built and nutrition. He was
not anorexic and psychological evaluation did not reveal
any abnormality. He was non diabetic and there was no
history of any other chronic illness or any prior surgery.
He had one binge eating episode about 48 hours ago. Afterwards he developed the symptoms and was admitted
in a primary health centre from where the patient was
referred to our institution.
Ischemia is caused presumably due to venous insufficiency when massive dilatation occurs [11, 12]. To impair
venous return, either 14mmHg of pressure or more than
3 litres of fluid is sufficient, although more than 15 litres has been described in eating disorders in chronic
distension.
Rupture can occur with intragastric pressures of more
than 120mmHg or 4 litres of fluid. In the majority of the
cases, greater curvature and gastric fundus are more
prone for necrosis and require emergent treatment [13].
Lesser curvature and pyloric regions of the stomach tend
to be spared [1].
A consequence of events as postulated by Abdu et al. is
mucosal necrosis, followed by full-thickness involvement
of the gastric wall and perforation [10–12]. Surgery may be
avoided if the diagnosis is established in an early stage. A
mortality rate of 80% to 100% has been reported due to
gastric ischemia and perforation as a result of dilation [14].
In our case, the stomach was hugely dilated and the greater curvature was below the umbilicus going towards the
pelvis. The gastric wall was thinned out and few patchy
areas of discolouration seen. No definite area of full
thickness gastric wall necrosis seen. There was one area
of serosal damage with impending perforation seen on
the body of the stomach. Patient received naso gastric
suction at the primary health centre which we believe
although failed to relieve the patient, but prevented the
rise of intra-gastric pressure to very high level and causing full thickness necrosis of the stomach.
Several theories have been postulated to explain the
pathogenesis of acute gastric dilatation. Morris et al.
claimed that anaesthesia and debilitation may be predisposing factor as it is a very frequent postoperative complication. Relaxation of the upper oesophageal sphincter with aerophagia may be a factor leading to gastric
distention [3, 4, 10]. In 1859, Brinton introduced the atonic
theory [10]. The stomach undergoes atony and muscular
atrophy during a period of starvation, so that a sudden
ingestion of food overtaxes an already weakened stomach in patients with eating disorders. In 1861, von Rokitansky proposed superior mesenteric artery syndrome
(mechanical theory) in which vascular compression of
the third segment of the duodenum, between superior
mesenteric artery, aorta, and vertebral column, causes
acute gastric dilatation [5]. Other authors suggest that
pancreatitis, peptic ulcer, gallbladder disease, and appendicitis also cause acute gastric dilatation [15, 16] and
infectious causes like necrotizing gastritis generally
involving immunocompromised patients like diabetes,
AIDS, and neoplasia are also reported [17, 18].
In more than 90% of cases of acute gastric dilatation,
vomiting is an important and common symptom [19]. Another sign reported in the literature is the inability to
vomit which is not fully understood. This may be due
to the occlusion of the gastroesophageal junction by the
distended.
Fundus, which angulates the oesophagus against the
right crus of the diaphragm, producing a one-way valve
[20]. Significant, diffuse abdominal distension accompanied by abdominal pain is common.
Plain abdominal radiograph and CT scan can demonstrate gastric distension and free air if present. In this
patient plain abdominal radiographs revealed grossly
distended stomach but no free air. As the patient was
not improving with conservative measures and he was
clinically deteriorating, we opted for emergency laparotomy fearing imminent perforation or necrosis. Treatment focuses on early diagnosis and decompression of
the stomach, thus halting the vascular congestion and
thus ischemia [21].Decompression with nasogastric tube
should be the first step in the management, followed by
immediate surgery in case of perforation. A normal size
nasogastric tube may prove to be inefficient in decompressing stomach. Sometimes, when semisolid material
is present in the stomach, even a large tube may be inefficient. In our case too since the contents were semisolid nasogastric tube was non-productive. If conservative
measures fail or gastric infarction with or without perforation is suspected, immediate surgical intervention
is mandatory [10].
We performed a gastrotomy for proper decompression
of the stomach near the greater curvature at the most dependent part and after decompressing, we performed a
side to side, two layered gastrojejunostomy. Primary repair done of the small area of serosal damage. We believe
that, in this condition a drainage procedure was better
than simple closure of the gastrotomy wound in view of
preventing recurrence. It has been reported to perform
partial or even total gastrectomy depending on the area
of the necrosis and general condition of the patient.
Surgeons should be aware that acute gastric dilatation
may occur even in patients who are not diagnosed as
having a typical eating disorder after an unaccustomed
episode of binge eating. A high index of suspicion is
necessary to diagnose this condition in order to avoid fatal complications. First line of treatment should be
conservative with nasogastric decompression. If it fails,
necessary timely surgery would prevent unnecessary
morbidity.
Declaration
Conflicts of interest
Te authors declare that there is no conflict of interest among the authors regarding publication of this paper.
References
1. Todd, S. R., Marshall, G. T., & Tyroch, A. H. (2000). Acute
gastric dilatation revisited. The American Surgeon, 66(8),
709.
2. T H SOMERVELL. (1945). Physiological gastrectomy. British Journal of Surgery, 33(1), 146-152.
3. Byrne, J. J., & Cahill, J. M. (1961). Acute gastric dilatation, 101(3), 301-309.
4. Cogbill, T. H., Bintz, M. A. R. I. L. U., Johnson, J. A., & Strutt,
P. J. (1987). Acute gastric dilatation after trauma. The Journal of trauma, 27(10), 1113-1117.
5. Adson, D. E., Mitchell, J. E., & Trenkner, S. W. (1997). The
superior mesenteric artery syndrome and acute gastric
dilatation in eating disorders: a report of two cases and
a review of the literature. International Journal of Eating
Disorders, 21(2), 103-114.
6. Turan, M., Şen, M., Canbay, E., Karadayi, K., & Yildiz, E.
(2003). Gastric necrosis and perforation caused by acute
gastric dilatation: report of a case. Surgery today, 33(4),
302-304.
7. Jefferiss, C. D. (1972). Spontaneous rupture of the stomach in an adult. British Journal of Surgery, 59(1), 79-80.
8. Wharton, R. H., Wang, T., Graeme-Cook, F., Briggs, S., &
Cole, R. E. (1997). Acute idiopathic gastric dilatation
with gastric necrosis in individuals with Prader-Willi
syndrome. American journal of medical genetics, 73(4),
437-441.
9. Evans, D. S. (1968). Acute dilatation and spontaneous rupture of the stomach. British Journal of Surgery, 55(12),
940-942.
10. Abdu, R. A., Garritano, D., & Culver, O. (1987). Acute gastric necrosis in anorexia nervosa and bulimia: two case
reports. Archives of Surgery, 122(7), 830-832.
11. Mishima, T., Kohara, N., Tajima, Y., Maeda, J., Inoue, K., Ohno,
T., ... & Kuroki, T. (2012). Gastric rupture with necrosis following acute gastric dilatation: report of a case. Surgery
today, 42(10), 997-1000.
12. Lunca, S., Rikkers, A., & Stãnescu, A. (2005). Acute massive
gastric dilatation: severe ischemia and gastric necrosis
without perforation. Rom J Gastroenterol, 14(3), 279-283.
13. Kerstein, M. D., Goldberg, B., Panter, B., Tilson, M. D.,
& Spiro, H. (1974). Gastric infarction. Gastroenterology, 67(6), 1238-1239.
14. Koyazounda, A., LeBaron, J. C., Abed, N., Daussy, D., Lafarie,
M., & Pinsard, M. (1985). Gastric necrosis from acute gastric dilatation: recovery after total gastrectomy. Journal de
Chirurgie, 122(6-7), 403-407.
15. Backett, S. A. (1985). Acute pancreatitis and gastric dilatation in a patient with anorexia nervosa. Postgraduate
Medical Journal, 61(711), 39-40.
16. Gondos, B. (1977). Duodenal compression defect and
the “superior mesenteric artery syndrome”. Radiology, 123(3), 575-580.
17. Nagai, T., Yokoo, M., Tomizawa, T., & MORI, M. (2001). Acute
gastric dilatation accompanied by diabetes mellitus. Internal Medicine, 40(4), 320-323.
18. Le Scanff, J., Mohammedi, I., Thiebaut, A., Martin, O., Argaud, L., & Robert, D. (2006). Necrotizing gastritis due to
Bacillus cereus in an immunocompromised patient. Infection, 34(2), 98-99.
19. Chaun, H. U. G. H. (1969). Massive gastric dilatation of
uncertain etiology. Canadian Medical Association journal, 100(7), 346.
20. Breslow, M., Yates, A., & Shisslak, C. (1986). Spontaneous
rupture of the stomach: A complication of bulimia. International Journal of Eating Disorders, 5(1), 137-142.
21. Baldassarre, E., Capuano, G., Valenti, G., Maggi, P., Conforti,
A., & Porta, I. P. (2006). A case of massive gastric necrosis
in a young girl with Rett Syndrome. Brain and development, 28(1), 49-51.