Open Access | Case Report
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Solitary fibrous tumor of the pleura: A rare mesenchymal tumor presented with hypoglycemia
*Corresponding author: Yasser Ali Kamal
Mailing address: Department of Cardiothoracic Surgery, Faculty
of Medicine, Minia University, El-Minia, 61111, Egypt.
Email: yaser_ali_kamal@yahoo.com, Yasser.Ali@mu.edu.eg
Received: 09 December 2020 / Accepted: 23 February 2021
DOI: 10.31491/CSRC.2021.03.070
Abstract
Solitary fibrous tumor (SFT) is a relatively rare primary tumor of the pleura, originating from the mesenchymal cells. The tumor has a non-specific clinical, laboratory, and radiological features. Patients may be asymptomatic or have non-specific clinical features including chest pain, dyspnea, cough, and paraneoplastic symptoms. Complete surgical resection is indicated in all cases to avoid recurrence of the tumor. We described a 49-year-old woman who presented with unexplained right chest pain and repeated attacks of hypoglycemia, associated with pleural SFT.
Keywords
Pleura; thoracic oncology; solitary fibrous tumor; paraneoplastic hypoglycemia; Doege-Potter syndrome
Introduction
The primary tumors of the pleura are less common
than secondary metastatic ones. Malignant mesothelioma is the most common primary tumor; however, other
rare pleural tumors exist including solitary fibrous tumor (SFT), lipoma, lymphoma, sarcoma, Askin tumor, extra-skeletal osteosarcoma, and histiocytoma [1].
SFT of the pleura is a relatively rare mesenchymal tumor that had earlier names of localized mesothelioma and pleural fibroma as it was thought to have a mesothelial origin. Most of the cases of SFT occur within the pleura, but it can arise from other sites including subcutaneous tissues, orbit, salivary glands, thyroid gland, nasal cavity, nasopharynx, lung, pericardium, adrenal
glands, liver, GI tract, urinary bladder, and prostate [2,3]. Here, we describe a case of SFT of the pleura with mild right chest pain and unexplained hypoglycemia.
Case Report
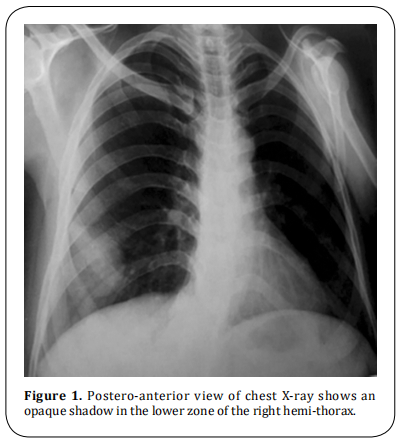
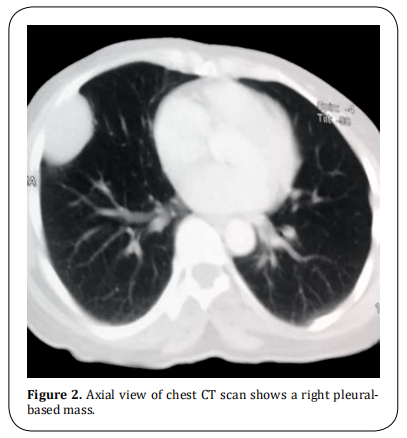
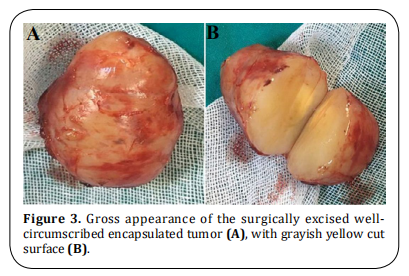
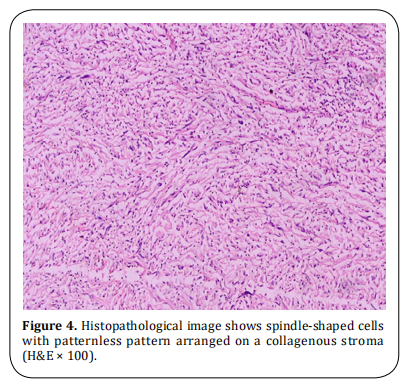
A 49-year-old previously healthy woman presented to our clinic with a 3-month history of mild right chest pain and repeated attacks of confusion and drowsiness improved after eating. There was laboratory evidence of hypoglycemia during the last attack (blood glucose level of 40 mg/dl). Chest X-ray showed an opaque shadow on the lower zone of the right side of the chest (Figure 1). The past medical history was unremarkable with normal laboratory investigations. A computed tomography (CT) scan revealed a well-defined right pleural-based soft tissue mass with a long axis diameter of 10 cm (Figure 2). CT-guided core needle biopsy revealed a spindle cell tumor. The patient underwent the right posterolateral thoracotomy. During surgery, the tumor was located in the right inferior hemithorax within the fifth and sixth intercostal spaces and it was attached to the parietal pleura by a fibrous pedicle. The tumor was completely resected. Grossly, the resected mass was round firm, well circumscribed encapsulated of 4.5×3 cm in diameter and the cut surface with grayish, yellow and mucoid (Figure 3). Microscopically, there was a bland spindle cell proliferation distributed in a patternless fashion separated by dense collagen stroma, with low mitotic figures less than 3/10 HPFs (Figure 4). On immunohistochemical examination, the tumor was positive for cluster of differentiation (CD- 34), CD99, and Vimentin and negative for S-100. The final diagnosis was benign pleural SFT. The postoperative course was uneventful, with freedom from symptoms and tumor recurrence within two years of follow-up after surgery.
Discussion
SFT is a mesenchymal cell tumor most commonly
occurs in middle-aged adults, with an equal gender
distribution [4]. The thoracic SFT occurs equally on
the right and left sides and arises from the pleura in most cases, followed by lung, anterior mediastinum,
and diaphragm [5]. The etiology of SFT remains unclear
with the absence of association to smoking or asbestos
exposure [6]. However, there is a genetic evidence recognizing SFT as a translocation-associated neoplasm,
with a consistent association with NAB2-STAT6 gene
fusions arising from recurrent intrachromosomal rearrangements on chromosome 12q [7].
Clinically, SFT presented as a slowly growing painless
mass that may produce symptoms due to its large size
or pressure effects on the adjacent structures. More
than 50% of cases with SFT are asymptomatic and detected incidentally on chest radiographs or CT. Symptomatic SFT presented with non-specific complaints
such as pleuritic chest pain, cough, and shortness of
breath [8]. The CT features are often quite suggestive
but not pathognomonic. On CT scan with contrast, SFT
appears as a heterogeneous tumor with acute angles
at pleural interface for large masses and obtuse angles
for smaller tumors [9]. A biopsy is recommended before starting treatment of SFT through open incisional
biopsy or by a core needle with a preference for radiological guided biopsy if it is applicable.
Histologically, SFT is a benign tumor although malignant transformation occurs in 20% of patients [10].
Also, SFT has a wide differential diagnosis on the histological examination due to the similarity in cellular
architecture and vascular pattern with other tumors
such as schwannoma, spindle cell/pleomorphic lipomas, gastrointestinal stromal tumors, and liposarcoma
[7]. Immunohistochemistry is useful for the diagnosis
and differentiation of SFT from other spindle cell mesenchymal tumors. The most important and valuable
immunohistochemical positive markers in SFT are
CD34, CD99, Vimentin, Bcl-2 and STAT-6. Additionally,
some markers are negative in cases of SFT according to
the tumor location, such as S100, smooth muscle actin
(SMA), desmin, and calretinin [11].
Surgery is the gold standard treatment for all types of
SFT, as its benefit outweigh the risks and the prognosis
for malignant SFT is favorable with surgical resection
[10]. Chemotherapy and consecutive selective embolization of the feeding arteries are alternative options
when the SFT is non-resectable or metastatic [12]. The
goal of the surgical management of SFT relies on wide
resection margins and preservation of the surrounding
structures. Complete resection with adequate negative
margins is essential to reduce the rates of local recurrence and progression to metastatic disease [13,14].
SFT is usually cured after complete surgical resection; however, a recent systematic review found that
SFTs recurred after 5 years of surgery in at least 23%
of cases and the authors of the review recommended
a longer follow up schedule [15]. Moreover, the local
recurrence has been reported in about 10% of benign
SFT [16] and in more than 60% of malignant SFTs, which
indicates surgical re-resection in cases with positive
surgical margins [17]. The possibility of repeated recurrence after surgery indicates extended surgical resection if the pleural SFT invades the chest wall, lung,
diaphragm, or pericardium [18].
Our case of pleural SFT presented with paraneoplastic hypoglycemia, which is known as Doege–Potter
syndrome (DPS). This rare entity occurs due to inappropriate secretion of a high molecular-weight form
of insulin-like growth factor type II (IGF-II). This big
IGF-II activates insulin receptors, inhibits hepatic gluconeogenesis, and increases glucose uptake with subsequent hypoglycemia [12]. Thus, DPS is characterized
by non-islet cell hypoinsulinemic hypoglycemia, which may be life-threatening when food intake decreased [19].
It is important to consider DPS in a patient with SFT
and hypoglycemia, especially when the tumor size is
larger than 8 cm which is thought to be a risk factor for
this syndrome [20]. The diagnosis of DPS is based on the
clinical presentation of hypoglycaemic episodes with
SFT in absence of alternative causes or on laboratory
findings of increased IGF-II to IGF-I ratio and hypoinsulinemia during attacks of hypoglycemia [21]. Treatment of DPS associated with SFT should initially aim
to normalize the blood glucose level by high-glucose
diet and continuous intravenous infusion of 50% glucose (up to 50 g/hr), followed by tumor size reduction
through surgical resection, chemotherapy, radiotherapy, brachytherapy, and photodynamic therapy. However, complete surgical resection of SFT is the curative
therapy for DPS [22].
Conclusion
SFT is a relatively rare soft tissue tumor that often originates from the mesenchymal cells of the pleura. The diagnosis of SFT is difficult due to the absence of symptoms in most of the cases, in addition to nonspecific clinical, laboratory, and radiological features. Some cases may present with paraneoplastic hypoglycemia, known as Doege–Potter syndrome. Surgical resection is the mainstay in the treatment of SFT with or without hypoglycemia. Complete surgical resection is essential to avoid the recurrence of both benign and malignant tumors.
Declarations
Authors’ contributions
The author contributed solely to the article.
Conflicts of interest
The author declared that there are no conflicts of interest.
Ethics approval
Not applicable.
Consent for publication
The consent was obtained from the patient.
References
1. Sureka, B., Thukral, B. B., Mittal, M. K., Mittal, A., & Sinha,
M. (2013). Radiological review of pleural tumors. The
Indian journal of radiology & imaging, 23(4), 313.
2. Brunnemann, R. B., Ro, J. Y., Ordonez, N. G., Mooney, J., El-Naggar, A. K., & Ayala, A. G. (1999). Extrapleural solitary fibrous tumor: a clinicopathologic study of 24 cases.
Modern pathology: an official journal of the United States
and Canadian Academy of Pathology, Inc, 12(11), 1034-
1042.
3. Agarwal, V. K., Plotkin, B. E., Dumani, D., French, S. W.,
Becker, R., & Lee, P. (2009). Solitary fibrous tumor of
pleura: a case report and review of clinical, radiographic
and histologic findings. Journal of radiology case reports,
3(5), 16.
4. Demicco, E. G., Park, M. S., Araujo, D. M., Fox, P. S., Bassett, R. L., Pollock, R. E., ... & Wang, W. L. (2012). Solitary
fibrous tumor: a clinicopathological study of 110 cases
and proposed risk assessment model. Modern Pathology,
25(9), 1298-1306.
5. Gold, J. S., Antonescu, C. R., Hajdu, C., Ferrone, C. R., Hussain, M., Lewis, J. J., ... & Coit, D. G. (2002). Clinicopathologic correlates of solitary fibrous tumors. Cancer, 94(4),
1057-1068.
6. Davanzo, B., Emerson, R. E., Lisy, M., Koniaris, L. G., &
Kays, J. K. (2018). Solitary fibrous tumor. Translational
gastroenterology and hepatology, 3, 94.
7. Thway, K., Ng, W., Noujaim, J., Jones, R. L., & Fisher, C.
(2016). The current status of solitary fibrous tumor:
diagnostic features, variants, and genetics. International
Journal of Surgical Pathology, 24(4), 281-292.
8. Hohenforst-Schmidt, W., Grapatsas, K., Dahm, M., Zarogoulidis, P., Leivaditis, V., Kotoulas, C., ... & Kosan, B.
(2017). Solitary fibrous tumor: A center’s experience and
an overview of the symptomatology, the diagnostic and
therapeutic procedures of this rare tumor. Respiratory
medicine case reports, 21, 99-104.
9. Salahudeen, H. M., Hoey, E. T. D., Robertson, R. J., & Darby,
M. J. (2009). CT appearances of pleural tumours. Clinical
radiology, 64(9), 918-930.
10. Supakul, R., Sodhi, A., Tamashiro, C. Y., Azmi, S. S., &
Kadaria, D. (2015). Solitary fibrous tumor of the pleura: A
Rare cause of pleural mass. The American journal of case
reports, 16, 854.
11. Geramizadeh, B., Marzban, M., & Churg, A. (2016). Role
of immunohistochemistry in the diagnosis of solitary
fibrous tumor, a review. Iranian journal of pathology,
11(3), 195.
12. Han, G., Zhang, Z., Shen, X., Wang, K., Zhao, Y., He, J., ... &Liu, X. (2017). Doege–Potter syndrome: a review of the
literature including a new case report. Medicine, 96(27),
e7417.
13. Sung, S. H., Chang, J. W., Kim, J., Lee, K. S., Han, J., & Park, S.
I. (2005). Solitary fibrous tumors of the pleura: surgical
outcome and clinical course. The Annals of thoracic surgery, 79(1), 303-307.
14. Kayani, B., Sharma, A., Sewell, M. D., Platinum, J., Olivier,
A., Briggs, T. W., & Eastwood, D. M. (2018). A review of the
surgical management of extrathoracic solitary fibrous tumors. American journal of clinical oncology, 41(7), 687-
694.
15. Mercer, R. M., Wigston, C., Banka, R., Cardillo, G., Benamore, R., Nicholson, A. G., ... & Rahman, N. M. (2020).
Management of solitary fibrous tumours of the pleura: a
systematic review and meta-analysis. ERJ open research,
6(3), 0005-2020.
16. Harrison–Phipps, K. M., Nichols, F. C., Schleck, C. D., Deschamps, C., Cassivi, S. D., Schipper, P. H., ... & Pairolero, P.
C. (2009). Solitary fibrous tumors of the pleura: results of
surgical treatment and long-term prognosis. The Journal
of thoracic and cardiovascular surgery, 138(1), 19-25.
17. Robinson, L. A. (2006). Solitary fibrous tumor of the
pleura. Cancer control, 13(4), 264-269.
18. Forster, C., Roumy, A., & Gonzalez, M. (2019). Solitary fibrous tumor of the pleura with Doege–Potter syndrome:
Second recurrence in a 93-year-old female. SAGE open
medical case reports, 7, 2050313X18823468.
19. Schutt, R. C., Gordon, T. A., Bhabhra, R., Cathro, H. P., Cook,
S. L., McCartney, C. R., & Weiss, G. R. (2013). Doege-Potter
syndrome presenting with hypoinsulinemic hypoglycemia in a patient with a malignant extrapleural solitary fibrous tumor: a case report. Journal of medical case reports, 7(1), 1-7.
20. Zafar, H., Takimoto, C. H., & Weiss, G. (2003). DoegePotter syndrome. Medical Oncology, 20(4), 403-407.
21. Ahluwalia, N., Attia, R., Green, A., Cane, P., & Routledge, T.
(2015). Doege–Potter syndrome. The Annals of The Royal
College of Surgeons of England, 97(7), e105-e107.
22. Jang, J. G., Chung, J. H., Hong, K. S., Ahn, J. H., Lee, J. Y., Jo, J.
H., ... & Lee, J. K. (2015). A case of solitary fibrous pleura
tumor associated with severe hypoglycemia: doegepotter syndrome. Tuberculosis and respiratory diseases,
78(2), 120.