Open Access | Letter to Editor
This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Optimal induction treatment regimens for extranodal NK/T-cell lymphoma: lessons learned, challenges, and proposals
* Corresponding author: Liang Wang
Mailing address: Department of Hematology, Beijing Tongren
Hospital, Capital Medical University, Beijing 100730, China.
E-mail: wangliangtrhos@126.com
Received: 23 November 2020 / Accepted: 16 December 2020
DOI: 10.31491/APT.2020.12.045
Extranodal NK/T-cell lymphoma (ENKTCL) is a highly
aggressive and Epstein-Bar virus (EBV)-associated hematologic malignancy, most prevalent in the Far East and
South America [1]. No standard treatment strategy has
been defined. It is well recognized that combination of
asparaginase-based chemotherapy (CT) and radiotherapy
(RT) benefit early-stage ENKTCL. It has been validated
that upfront RT can cure most stage I patients of ENKTCL
without risk factors, and asparaginase (either L-asparaginase or pegaspargase) is the backbone of chemotherapy
for ENKTCL. In recent years, a variety of asparaginasebased regimens have been found to be effective in earlystage ENKTCL, such as GELOX (gemcitabine, oxaliplatin, and asparaginase) [2] or EPOCHL (etoposide,
prednisone, vincristine, cyclophosphamide, doxorubicin,
and asparaginase) [3], but with different toxicities. Thus,
we are wondering how much is too much for early-stage
ENKTCL patients, and is intensive chemotherapy really
necessary? Moreover, the international T-cell Project reported the updated survival outcomes of a large cohort of
worldwide patients with ENKTCL (except Chinese) this
year, and more than 60 regimens were used in this report,
indicating a lack of standard treatment regimens [4].
Compared with the survival data of Chinese patients with
ENKTCL, the outcomes reported by this international
multi-center cohort seem unsatisfactory. Due to very small
number of patients enrolled from the USA and no patients
from China, it is of importance to show the data from
these two large countries, which enable us to depict the
real-world data of ENKTCL globally. In this multi-center
Aging Pathobiology and Therapeutics 2020; 2(4):226-229 226
DOI: 10.31491/APT.2020.12.045
retrospective study, we compared the efficacy, long-term
survival outcomes, and safety profiles between GELOX
and EPOCHL regimen in early-stage ENKTCL patients.
Meanwhile, the survival outcomes of all-stage patients
from China and SEER (Surveillance, Epidemiology, and
End Results) database were compared.
A total of 494 patients of ENKTCL with all stages (AnnArbor stage I, n = 224; stage II, n = 168; stage III-IV,
n = 102) from Beijing Tongren Hospital were included
in this study, and all patients received asparaginasebased treatment, among whom 193 patients with stage
I-II ENKTCL were treated with 4-6 cycles of GELOX
(pegaspargase 2,500 IU/m2
d1, gemcitabine 1,000 mg/m2
d1, 8, oxaliplatin 100 mg/m2
d1, repeated every 3 weeks)
or EPOCHL (etoposide 50 mg/m2
/d, vincristine 0.5 mg/d,
doxorubicin 10 mg/m2
/d were administered by continuous
intravenous infusion over 96 h d1-4, prednisone 60 mg/
m2
/d was administered orally on days 1-5, cyclophosphamide 750 mg/m2
was given as an intravenous bolus on
day 5, L-asparaginase 10,000 U/d was administered by
intravenous infusion on days 6-10 or pegaspargase 2500
IU/m2
d1, repeated every 3 weeks) regimen combined
with radiotherapy (≥50Gy), either sequential or sandwich
chemoradiation. Response to treatment was evaluated using MRI scan or PET-CT scan after chemotherapy and end
of treatment. Progression free survival (PFS) was calculated from diagnosis of ENKTCL to disease progression,
death of any reason, or last follow-up, whichever came
first. Overall survival (OS) was defined from diagnosis to
death of any reason, or last follow-up, whichever came
first. The institutional ethics review board approved this
study and all patients provided informed consent for publication of clinical data at their first visit, but all patients
in this study had been de-identified. Moreover, we identified 1140 patients with ENKTCL from the SEER database
(www.seer.cancer.gov) based on the ICD-O-3 code for
histology (9719, ENKTCL) from 1987 to 2015, and 330
patients were excluded from our analysis due to unknown
stage or missing detailed survival data.
68 patients with early stage ENKTCL received GELOX
induction therapy and 125 patients were treated with EPOCHL regimen. No significant difference was found
between those two groups concerning baseline characteristics. At the end of treatment, a complete response (CR)
rate of 75% and an overall response rate (ORR) of 98.5%
was achieved in the GELOX group, and the CR rate and
ORR were 56% and 94.4% in the EPOCHL group, respectively. Although there was no significant difference
in ORR between GELOX and EPOCHL regimen, as is
shown in Figure 1, patients treated with GELOX had significantly better PFS and OS than those with EPOCHL
(p<0.05). Meanwhile, the safety profile of GELOX regimen was more favorable than EPOCHL, especially the
hematology toxicities. As expected, patients with stage II
disease had significantly inferior outcomes than those with
stage I disease (p < 0.05). The end of treatment efficacy
had a significant impact on the long-term survival outcomes, as patients with CR enjoyed more favorable PFS
and OS than those without CR (p<0.05). In the GELOX
cohort, efficacy assessment data was available before RT
consolidation. As is shown in Figure 1-H, patients without
CR after induction chemotherapy had significantly inferior PFS than those with CR (p = 0.033), though part of
those patients could be salvaged by RT consolidation and
attained CR at the end of RT.
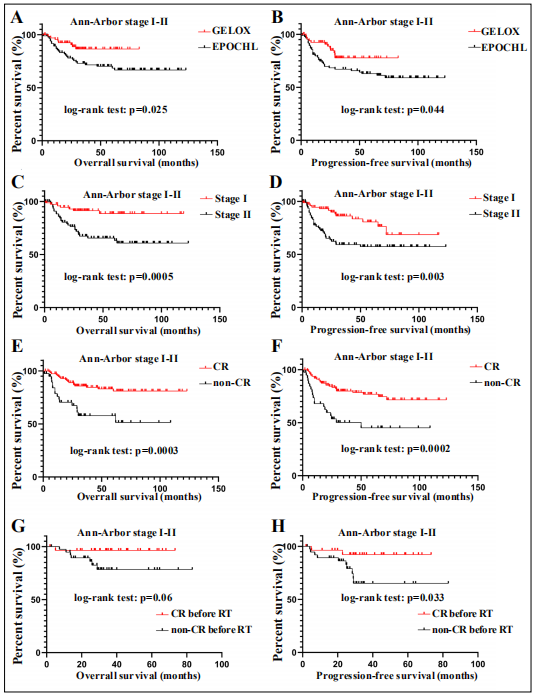
Figure 1. Survival curves for ENKTCL patients with localized disease. Patients treated with GELOX regimen had significantly better OS (A) and PFS (B) than those treated with EPOCHL. Patients with stage II disease h a d s i g n i f i c a n t l y inferior OS (C) and PFS (D) than those with stage I. The end of treatment response significantly correlated w i t h p r o g n o s i s o f ENKTCL (E, F). For patients treated with GELOX, CR before radiotherapy indicated significantly better PFS (H) and a trend for better OS (G). CR means complete response, RT means radiotherapy.
The detailed baseline characteristics of both Chinese and SEER cohorts were shown in Table 1. More patients in the SEER database were treated before 2010 when asparaginase-based therapies were not well recognized. Moreover, significantly more patients in the SEER database had extra-nasal or non-upper aerodigestive tract (UADT) disease. The treatment strategies were unknown for the SEER database, and asparaginase-based chemotherapy with or without combined radiotherapy was administrated in the Chinese cohort. As is shown in Figure 2, the fiveyear OS rate was 79.4%, 56.6%, and 43.6% for patients with stage I, II, and III-IV, respectively, in the Chinese cohort, which seemed much better than that in the SEER database (55.2%, 35.6%, and 21.2%, respectively). This discrepancy may be caused by unbalanced baseline characteristics and different treatment strategies. Also, the etiology and biology of ENKTCL may differ between Asian and Western countries.
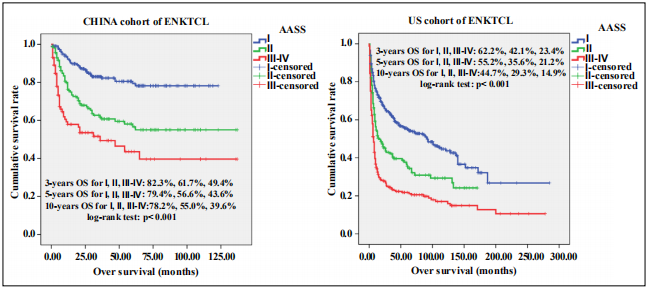
Figure 2. Survival curves for patients of ENKTCL in the Chinese (A) and SEER (B) cohorts. AASS means Ann-Arbor staging system.
Table1
The baseline characteristics of ENKTCL patients.
| Chinese cohort (N= 494) |
SEER database (N=241) |
P value | |
|---|---|---|---|
| Year of diagnosis | |||
| 1987-2009 2010-now |
191 (38.7%) 303 (61.3%) |
443 (54.7%) 367 (45.3%) |
P<0.001 |
| Gender | |||
| Male Female |
335 (67.8%) 159 (32.2%) |
524 (64.7%) 286 (35.3%) |
P=0.274 |
| Age | |||
| <60 ≥60 |
403 91 (18.4%) |
513 (63.3%) 297 (36.7%) |
P<0.001 |
| Ann-Arbor stage | |||
| I II III-IV |
179 (36.2%) 197 (39.9%) 118 (23.9%) |
381 (47.0%) 179 (22.1%) 250 (30.9%) |
P<0.001 |
| Primary lymphoma site | |||
| UADT Non-UADT Nasal cavity Extra-nasal sites |
452 (91.5%) 42 (8.5%) 320 (64.8%) 174 (35.2%) |
596 (73.6%) 214 (26.4%) 387 (47.8%) 423 (52.2%) |
P<0.001 |
Due to the distinct geographic pattern of ENKTCL, many studies were done in Asian countries, and asparaginasebased induction therapies were well recognized, should be given in the first-line setting. Nowadays, risk-model guided therapy should be implemented in clinical practice [5]. For those localized diseases at low-risk, radiotherapy alone could achieve satisfactory results, and asparaginasebased chemotherapy should be added to radiotherapy for those with risk factors. The standard induction regimens were not defined yet, and our previous study demonstrated that pegaspargase monotherapy in combination with concurrent radiotherapy could attain 100% CR rate for early stage disease [6], which needs to be validated in phase 3 RCTs. Although the prognosis is much better for the Chinese cohort, patients with the advanced disease still fare poorly. Increasing evidence has shown the advantage of immunotherapy (PD-1/PD-L1 blockade) in relapsed/ refractory ENKTCL [7], and we guess the first-line use of immunotherapy in combination with asparaginasebased chemotherapy might further improve the prognosis of patients with advanced disease [8]. Recently, a novel molecular subtyping system was defined for ENKTCL, including TSIM, MB, and HEA subtypes, with both prognostic value and treatment guidance [9]. In the future, prospective clinical trials should be conducted incorporating the new risk models and molecular subtyping system, to further implement precision medicine in ENKTCL.
Declarations
Conflict of interest
The author declares no conflict of interest.
References
1. Wang L, Li L R, Zhang L, et al. The landscape of new drugs in extranodal NK/T-cell lymphoma. Cancer Treatment Reviews, 2020, 89.
2. Wang L, Wang Z, Chen X, et al. First-line combination of gemcitabine, oxaliplatin, and L-asparaginase (GELOX) followed by involved‐field radiation therapy for patients with stage IE/IIE extranodal natural killer/T-cell lymphoma. Cancer, 2013, 119(2): 348-355.
3. Wei L, Wang J, Ye J, et al. Long-term outcomes of patients treated with an EPOCHL regimen as first-line chemotherapy for newly diagnosed extranodal natural killer/ T-cell lymphoma: a retrospective single-center study. Leukemia & Lymphoma, 2020, 61(2): 337-343.
4. Fox C P, Civallero M, Ko Y H, et al. Survival outcomes of patients with extranodal natural-killer T-cell lymphoma: a prospective cohort study from the international T-cell Project. The Lancet Haematology, 2020.
5. Chen S Y, Yang Y, Qi S N, et al. Validation of nomogramrevised risk index and comparison with other models for extranodal nasal-type NK/T-cell lymphoma in the modern chemotherapy era: indication for prognostication and clinical decision-making. Leukemia, 2020: 1-13.
6. Wang H, Wang L, Li C, et al. Pegaspargase Combined with Concurrent Radiotherapy for Early-Stage Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type: A Two-Center Phase II Study. The Oncologist, 2020, 25(11): e1725.
7. Wang L, Wang J. PD-1 blockade in extranodal NK/T-cell lymphoma: who is in charge?. Leukemia, 2020: 1-2.
8. Wang L, Wang J. Extranodal natural-killer T-cell lymphoma: experience from China. The Lancet Haematology, 2020, 7(6): e441.
9. Xiong J, Cui B W, Wang N, et al. Genomic and Transcriptomic Characterization of Natural Killer T Cell Lymphoma. Cancer Cell, 2020, 37(3): 403-419. e6.