Open Access | Research
This work is licensed under a Creative
Commons Attribution-ShareAlike 4.0 International License.
Impact of oral vitamin D3 supplementation on proximal femur fracture incidence in the elderly population
# These authors contributed equally to this work.
* Corresponding author: Hao Liu
Mailing address: The Second Affiliated Hospital of Nanchang
University, Nanchang Jianyuan Rehabilitation Hospital, No. 1
Minde Road, East Lake District, Nanchang, 330006, Jiangxi,
China.
Email: liuhao952252@163.com
Received: 15 May 2024 / Revised: 03 June 2024 / Accepted: 11 June 2024 / Published: 27 June 2024
DOI: 10.31491/APT.2024.06.141
Abstract
Objective: To evaluate the effect of oral vitamin D3 supplementation on the incidence of proximal femur fracture (PFF) in the elderly population.
Materials and Methods: We searched PubMed and Embase databases for relevant studies from inception to March 2024. Only randomized controlled trials (RCTs) were included. Raw data were extracted into a predefined worksheet, and quality analysis of RCTs was performed using the risk-of-bias tool version 2 (RoB2).
Results: Thirteen RCTs were included in the meta-analysis. Oral vitamin D3 supplementation alone in the elderly did not show a significant association with PFF incidence (OR = 1.04, 95% CI: 0.94-1.15, p-value = 0.46 > 0.05). Combined oral supplementation of vitamin D3 and calcium could reduce the incidence of PFF in the elderly population (OR = 0.80, 95% CI: 0.69-0.94, p-value = 0.005 < 0.05); however, after excluding low-quality RCTs, the protective effect was no longer statistically significant (OR = 0.90, 95% CI: 0.74-1.10, p-value = 0.32 > 0.05).
Conclusion: Limited evidence suggests that combined oral supplementation of vitamin D3 and calcium may reduce the incidence of PFF in the elderly population, as the protective effect was no longer statistically significant after excluding low-quality RCTs.
Keywords
Vitamin D3, cholecalciferol, calcium, proximal femur fracture, elderly population, meta-analysis
Introduction
Osteoporosis is a pathological condition characterized by
loss of bone mass and structural deterioration, resulting
in increased fracture susceptibility [1]. Epidemiological
evidence indicates a significant prevalence of osteoporotic
fractures, with an estimated one in two women and one in
five men aged 65 years and older expected to experience
an osteoporotic fracture during the remainder of their lives
[1, 2]. Given the demographic trend toward an increasingly elderly population, prevention of osteoporotic fractures in the elderly is of paramount importance. Among
the fractures associated with osteoporosis, proximal femur
fracture (PFF), commonly known as hip fracture, has the
greatest impact on geriatric patients (≥ 65 years of age)
due to its high mortality rate, long-term disability, and loss
of independence [3, 4]. The documented one-year mortality after PFF is estimated to range from 14% to 58%, associated with a 4% annual increase in the relative risk of
mortality in geriatric patients [5]. In addition, the process
of hospitalization, rehabilitation and potential need for
long-term care places a significant socioeconomic burden
on the healthcare infrastructure. This underscores the critical need for tailored preventive interventions in geriatric
PFF patients.
Vitamin D3 (cholecalciferol) plays a critical role in maintaining musculoskeletal health by facilitating calcium
absorption, promoting osteoid tissue mineralization in
bone, and maintaining muscle function [6, 7]. Vitamin
D3 deficiency is commonly defined by experts as a serum
25-hydroxyvitamin D (25(OH)D) level below 20 ng/mL
(50 nmol/L), which can lead to secondary hyperparathyroidism, bone demineralization, and muscle weakness [8,
9]. Many previous studies have claimed that daily oral supplementation of vitamin D3 (including vitamin D3
alone and combined supplementation of vitamin D3 and
calcium) has the effect of preventing PFF and relieving
its symptoms [10, 11]. However, the efficacy of such interventions remains controversial, as evidenced by some
recent studies indicating that oral vitamin D3 supplementation is not significantly associated with a reduced risk of
PFF in geriatric patients [12-14]. Therefore, we conducted
a comprehensive meta-analysis based on relevant randomized controlled trials (RCTs) to evaluate the effect of oral
vitamin D3 supplementation (including vitamin D3 alone
and combined supplementation with vitamin D3 and calcium) on the incidence of PFF in the elderly population
(aged 65 years and older).
Materials and methods
Search strategies
A systematic literature search was conducted in March 2024 using PubMed and Embase databases. To increase the breadth of literature coverage, the reference lists of candidate studies were carefully reviewed by hand to ensure comprehensive access to relevant scientific materials.
Inclusion and exclusion criteria
Two reviewers performed independent assessments to determine eligibility of candidate articles. Articles deemed irrelevant after abstract review were excluded. All abstracts meeting the initial criteria were reviewed as full articles. All abstracts that met the initial criteria were reviewed as full articles. Non-English publications, animal studies, basic science studies, case reports, commentaries, letters, meeting abstracts, protocols, and review articles were excluded from consideration. Studies were selected using the following inclusion criteria: (1) RCTs comparing vitamin D3 alone or combined vitamin D3 and calcium oral supplements with placebo/no treatment; (2) studies that enrolled participants aged 65 years and older (or could extract data only from participants aged 65 years and older); (3) studies with at least 5 PFF events to reduce random error; (4) studies with at least 1 year of followup; (5) studies that provided access to raw data. In cases where different studies used overlapping data, the study with the more comprehensive statistics was preferred. Disagreements were resolved through consensus discussions, with the involvement of a third party when necessary.
Risk of bias assessment
Risk of bias was assessed using the Cochrane risk-of-bias
tool version 2 (RoB2), and visual representations were
generated using the R package "robvis" [15, 16]. RoB2,
which is tailored for RCTs, rigorously assesses each domain for its potential bias and categorizes it as low risk,
some concern, or high risk [16]. The RoB2 domains are:
• Bias due to the randomization process
• Bias due to deviations from the intended interventions
• Bias due to missing outcome data
• Bias in outcome measurement
• Bias in selection of reported outcome
Data analysis
The R package "meta" was used for the following statistical analysis [17]. The standardized mean difference (SMD) along with a 95% confidence interval (CI) was used as the statistic for effect analysis of the measures. The degree of heterogeneity was also determined by combining I2 quantification and Q statistic. If the I2 value exceeded 50% or the p-value of the Q statistic was less than 0.05, indicating substantial heterogeneity among the included studies, the random effects model was considered more appropriate. Conversely, if these thresholds were not met, the common effect model (also referred to as the fixed effect model) was preferred.
Sensitivity analysis
In addition, sensitivity analyses were performed to assess the impact of individual studies, particularly those of low quality, on the overall effect size estimate.
Results
Search results
A total of 467 articles were identified for the initial review. After comprehensive evaluation of the full texts, 13 RCTs met our criteria for further meta-analysis (Figure 1).
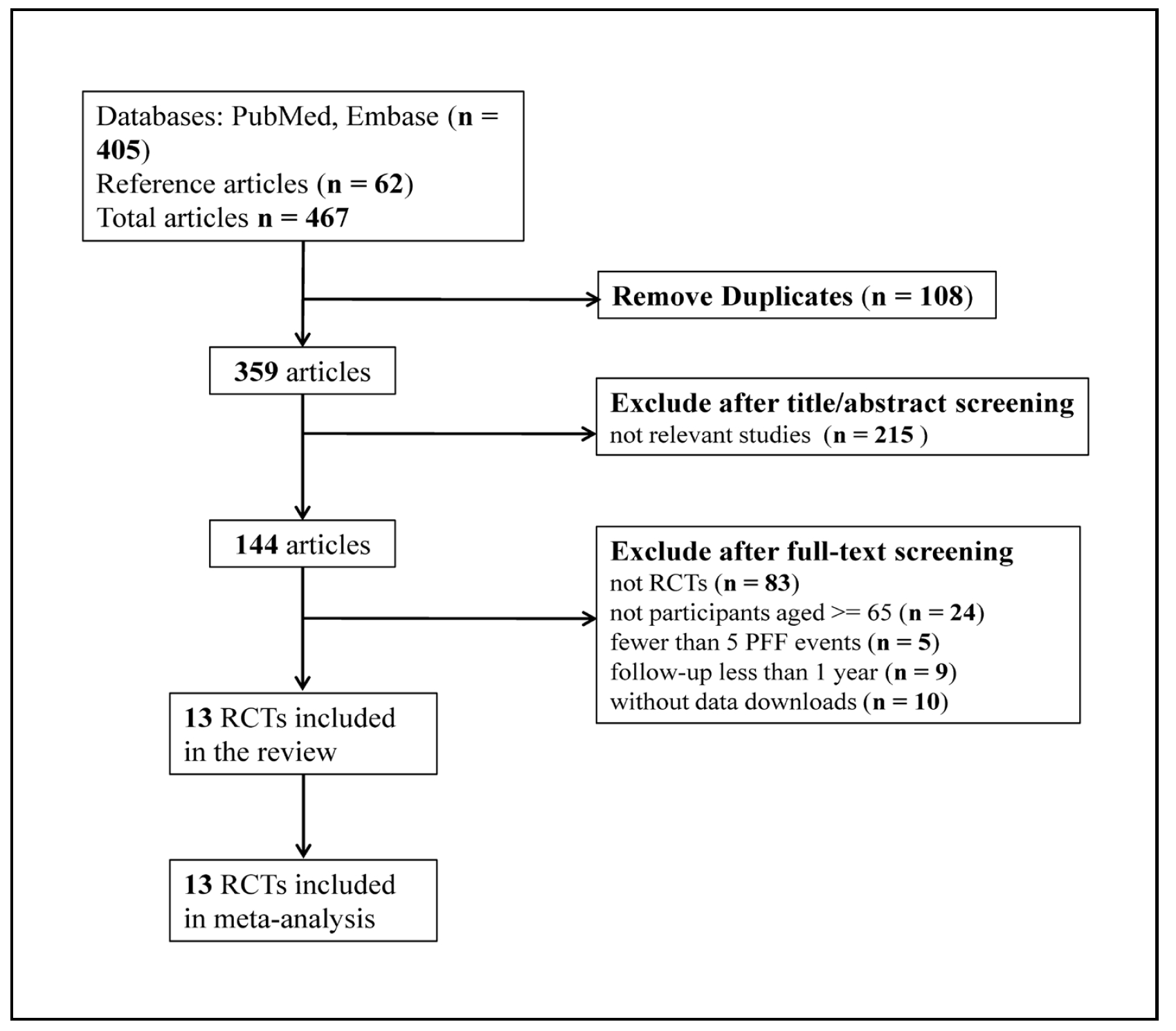
Figure 1. Flow chart of search strategy and study selection.
Specifically, 8 RCTs used a design that compared the incidence of PFF with oral vitamin D3 supplementation alone versus placebo/no treatment. Six RCTs used a design comparing the PFF incidence of combined oral supplementation with vitamin D3 and calcium versus placebo/ no treatment. Of note, 1 RCT included both of the above experimental designs, resulting in a total of 13 RCTs selected. The characteristics of all included RCTs are listed below ( Table 1 & 2).
Table 1
Characteristics of 8 RCTs comparing the incidence of PFF in participants aged 65 years or older between oral vitamin D3 supplementation
alone and placebo/no treatment.
| Study | Year | Country | Participants, No. | Treatment (vitamin D3 alone) | Mean Age, y | Follow-up, y | PFF, No. |
|---|---|---|---|---|---|---|---|
| Lips [18] | 1996 | Netherlands | 2578 | 400 IU/d | 80 | 3.5 | 106 |
| Meyer [19] | 2002 | Norway | 1144 | 400 IU/d | 84.7 | 2 | 97 |
| Trivedi [20] | 2003 | UK | 2686 | 100,000 IU/4 mo | 74.8 | 5 | 45 |
| Grant [21] | 2005 | UK | 2675 | 800 IU/d | 77 | 3.8 | 88 |
| Lyons [22] | 2007 | UK | 3440 | 100,000 IU/4 mo | 84 | 3 | 216 |
| Smith [23] | 2007 | UK | 9440 | 300,000 IU/y | 79.1 | 3 | 110 |
| Sanders [24] | 2010 | Australia | 2258 | 500,000 IU/y | 76.1 | 5 | 34 |
| Waterhouse [26] | 2023 | Australia | 15306 | 60,000 IU /mo | 65+ | 5 | 789 |
Table 2
Characteristics of 6 RCTs comparing the incidence of PFF in participants aged 65 years or older between combined oral vitamin D3 &
calcium supplementation and placebo/no treatment.
| Study | Year | Country | Participants, No. | Treatment | Mean Age, y | Follow-up, y | PFF, No. |
|---|---|---|---|---|---|---|---|
| Chapuy [25] | 1994 | France | 2303 | 800 IU/d vitamin D3, 1200 mg/d calcium | 84 | 3 | 322 |
| Chapuy [26] | 2002 | France | 583 | 800 IU/d vitamin D3, 1200 mg/d calcium | 85.2 | 2 | 105 |
| Grant [21] | 2005 | UK | 2638 | 800 IU/d vitamin D3, 1000 mg/d calcium | 77.5 | 5 | 87 |
| Porthouse [27] | 2005 | USA | 3314 | 800 IU/d vitamin D3, 1000 mg/d calcium | 76.8 | 2 | 25 |
| Jackson [28] | 2006 | USA | 6340 | 400 IU/d vitamin D3, 1000 mg/d calcium | 70+ | 7 | 208 |
| Salovaara [29] | 2010 | Finland | 3195 | 800 IU/d vitamin D3, 1000 mg/d calcium | 67.3 | 4.3 | 6 |
Assessment of risk of bias
The result of the RoB2 "risk of bias" assessment for the selected 13 RCTs is shown in Figure 2. Most studies were assessed as having a low or moderate risk of bias, and only 2 studies were assessed as having a high risk of bias in at least one domain.
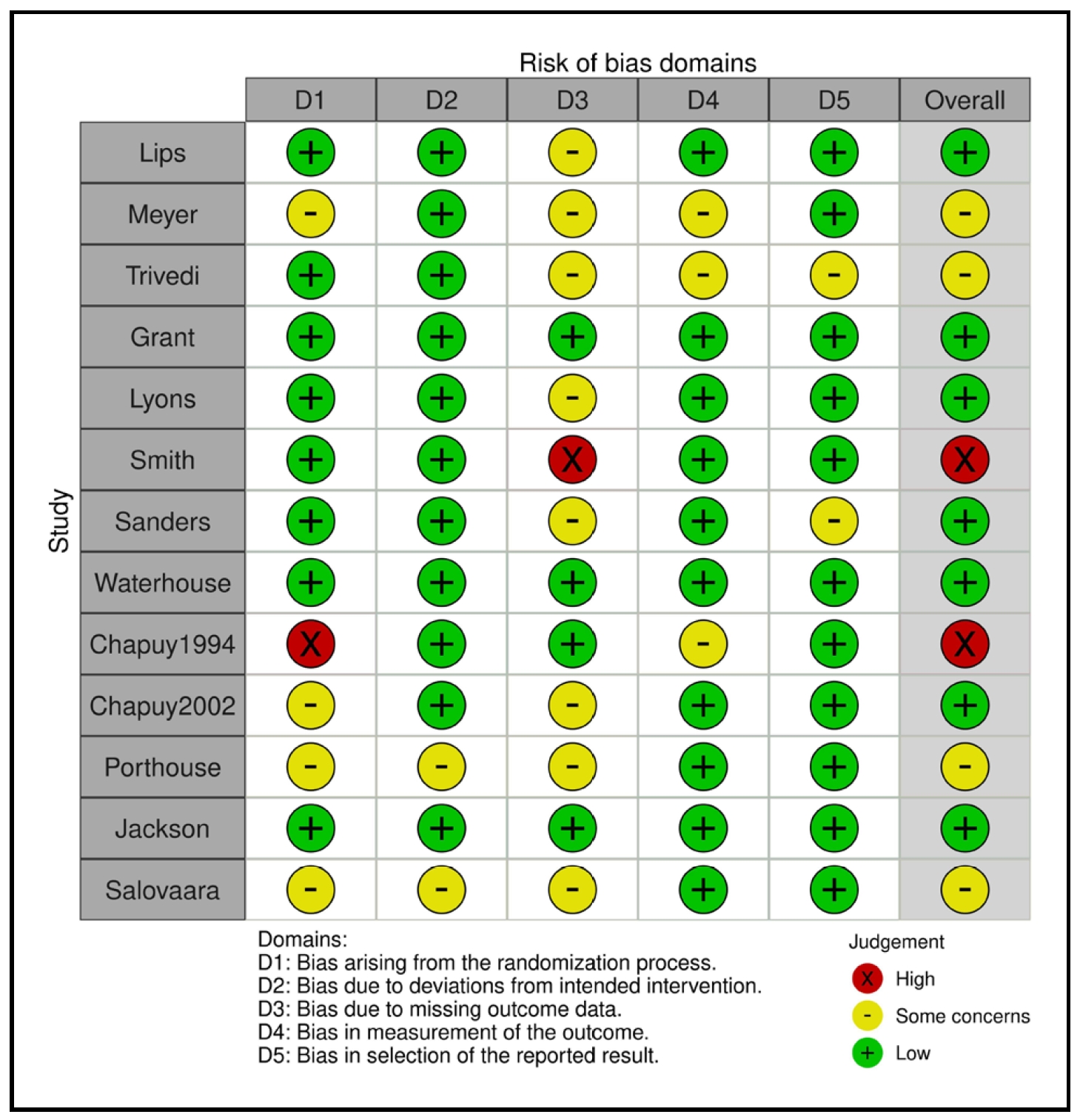
Figure 2. RoB2 risk of bias assessment of the selected 13 RCTs.
Oral vitamin D3 supplementation alone
A total of 8 RCTs evaluated the association between PFF
incidence and oral vitamin D3 supplementation alone.
Seven RCTs showed no statistically significant association
[12, 18-22, 24], while the remaining RCT suggested that
oral vitamin D3 supplementation alone may increase the
incidence of PFF [23].
No significant heterogeneity was found (Figure 3A, I2
=
0, Q-statistic p-value = 0.45 > 0.05); therefore, the fixedeffect model was used. The meta-analysis result showed
no significant association between PFF incidence and oral
vitamin D3 supplementation alone in the elderly population (Figure 3A, OR = 1.04, 95% CI: 0.94-1.15, p-value =
0.46 > 0.05).
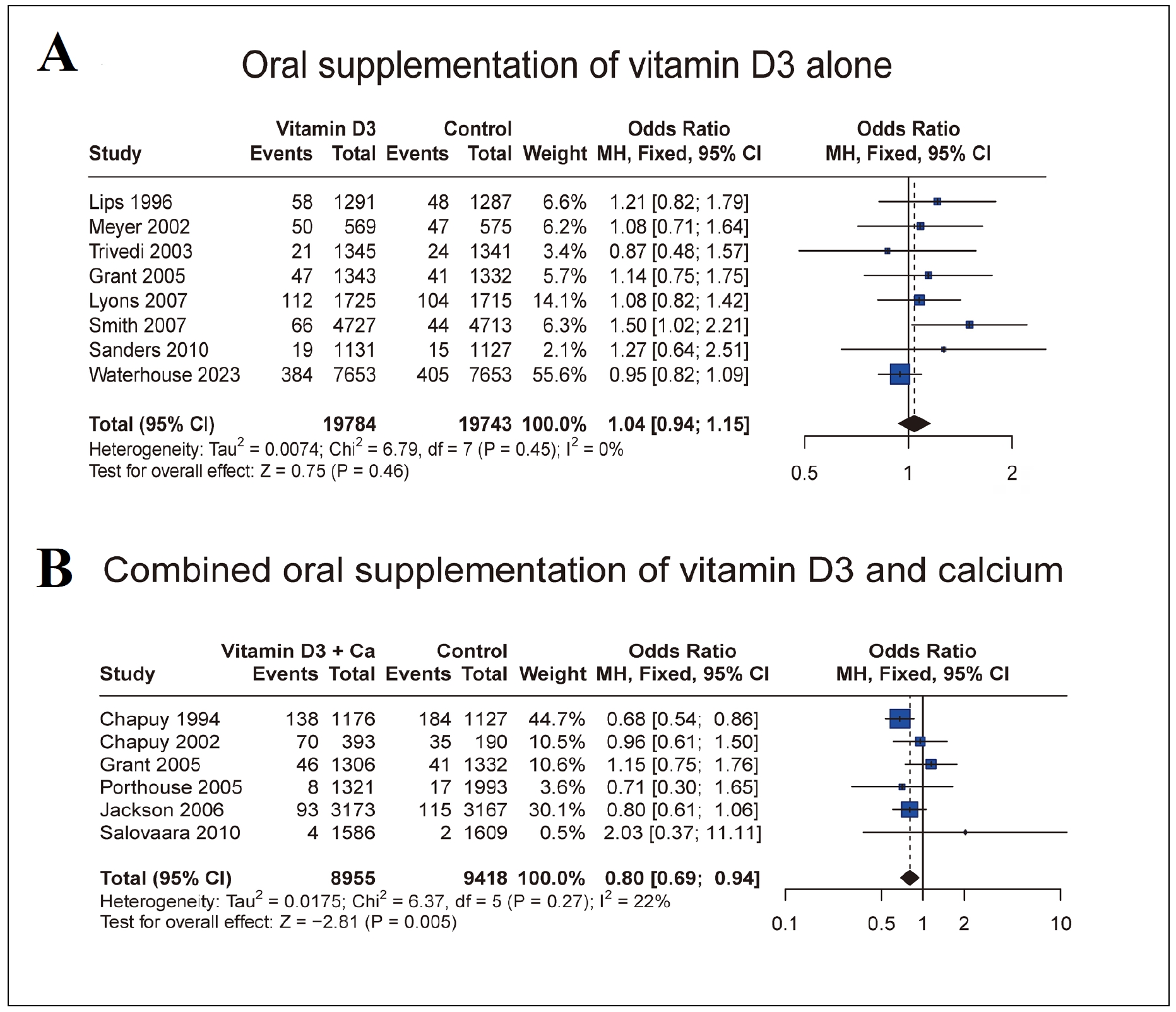
Figure 3. Forest plot for PFF incidence comparisons between oral vitamin D3 supplementation alone the placebo/no treatment (A) and between combined oral vitamin D3 supplementation + calcium and placebo/no treatment (B).
Combined oral vitamin D3 supplementation and calcium
Six RCTs evaluated the association between PFF incidence and combined oral supplementation with vitamin
D3 + calcium. Five RCTs showed no statistically signifcant association [21, 26-29], while the remaining RCT
suggested that combined oral supplementation with vitamin D3 + calcium could reduce the risk of PFF [25].
Since I2
= 22% and Q-statistic p-value = 0.27 > 0.05, we
performed fixed model (Figure 3B). The result indicated
that combined oral supplementation of vitamin D3 + calcium could significantly reduce the incidence of PFF in
the elderly population (Figure 3B, OR = 0.80, 95% CI:
0.69-0.94, p-value = 0.005 < 0.05).
Sensitivity analysis
The result of the sensitivity analysis showed that among the 8 RCTs of oral vitamin D3 supplementation alone, the exclusion of the studies deemed of low quality did not qualitatively change the original findings. When Smith's study was excluded, the lack of a significant association between PFF incidence and oral vitamin D3 supplementation alone in the elderly population persisted (Figure 4A, OR = 1.01, 95% CI: 0.91-1.13, p-value = 0.86 > 0.05). However, in the sensitivity analysis of 6 RCTs investigating combined oral supplementation with vitamin D3 + calcium, the exclusion of low-quality studies changed the original research results. Specifically, when the 1992 Chapuy study was excluded, we observed that combined oral supplementation with vitamin D3 and calcium no longer had an effect on the incidence of PFF in the elderly population (Figure 4B, OR = 0.90, 95% CI: 0.74-1.10, p-value = 0.32 > 0.05).
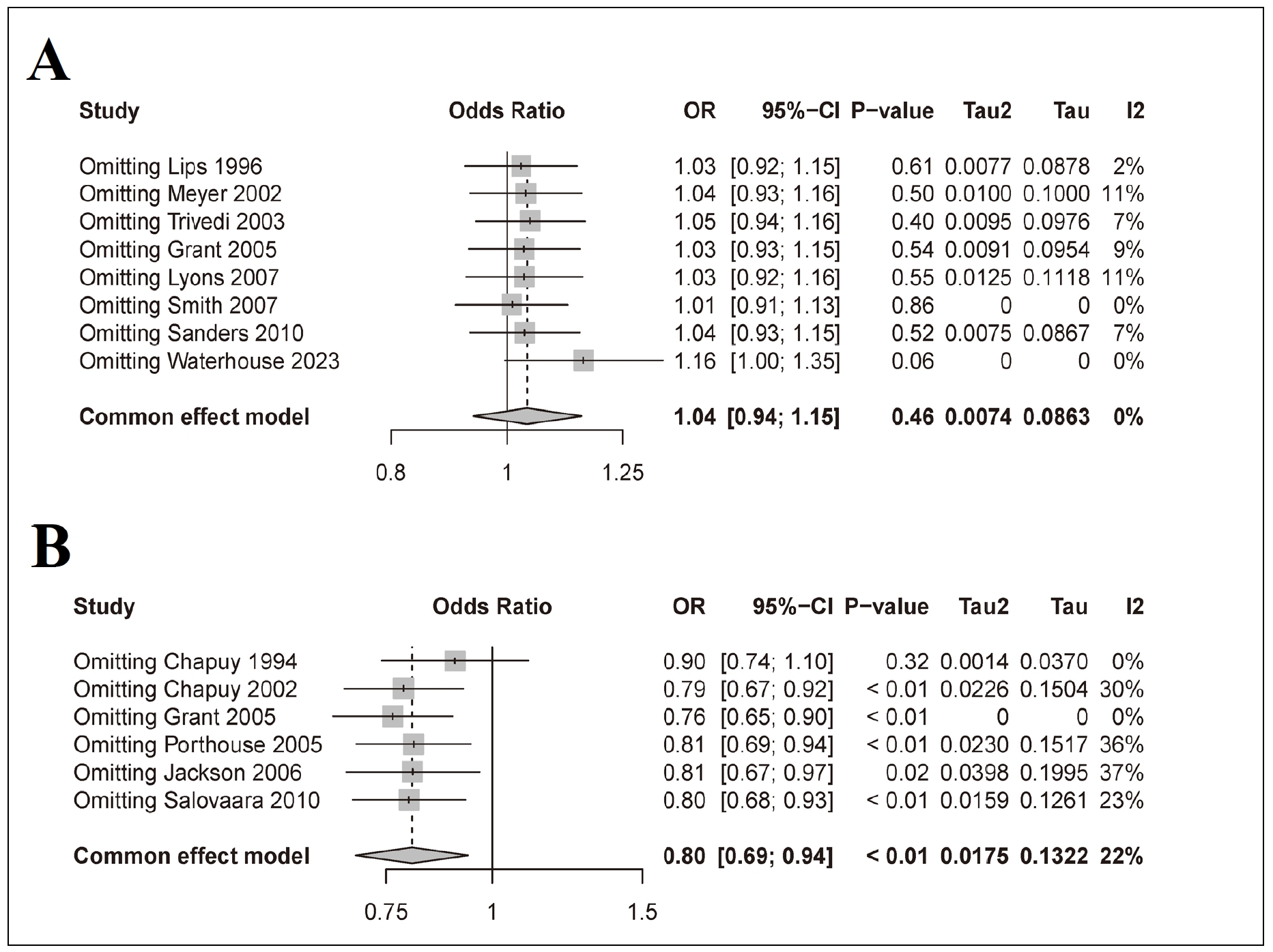
Figure 4. Sensitivity analysis of 8 RCTs designed for oral supplementation with vitamin D3 alone (A) and 6 RCTs designed for combined oral supplementation with vitamin D3 + calcium (B).
Discussion
The present meta-analysis, using rigorous inclusion criteria, included 13 RCTs with a total of 55,830 participants
to determine the efficacy of oral vitamin D3 supplementation in reducing the incidence of PFF in the geriatric
population. Our review shows that oral vitamin D3 alone
did not show a significant association with the incidence
of PFF in the elderly (OR = 1.04, 95% CI: 0.94-1.15, p-value = 0.46 > 0.05), a conclusion that remained consistent even after removing the study considered to be of low
quality (OR = 1.01, 95% CI: 0.91-1.13, p-value = 0.86 >
0.05). Meanwhile, when vitamin D3 was combined with
calcium and administered orally, a significant reduction
in PFF incidence was observed in the elderly (OR = 0.80,
95% CI: 0.69-0.94, p-value = 0.005 < 0.05). However, after excluding the study of inferior quality, this association
lost statistical significance (OR = 0.90, 95% CI: 0.74-1.10,
p-value = 0.32 > 0.05).
Bischoff-Ferrari et al. previously found a significant association between daily supplementation with vitamin
D3 alone and a lower incidence of PFF in adults aged 65
years or older; however, their previously published pooled
analysis suffered from limitations such as inadequate sample size and follow-up duration in some of the included
RCTs, which to some extent affected the generalizability
and statistical power of the final results [30]. At the same
time, Bischoff-Ferrari suggested that the efficacy of daily
oral vitamin D3 supplementation in reducing PFF incidence might be significant only at high doses (800 IU or
more per day). However, sensitivity analysis in our metaanalysis showed that this association remained statistically
insignificant even after excluding RCTs with doses below
800 IU per day [30].
The trials of combined oral supplementation with vitamin
D3 + calcium conducted by Avenell and Manoj produced
results that were consistent with the results of our metaanalysis, demonstrating that the intervention was effective
in reducing the incidence of PFF in the elderly population
[31, 32]. It is important to note that our meta-analysis
also highlights the influence of low-quality RCTs on the
results of the effect analysis. As in previous similar analyses, we included Chapuy's 1994 study [25]. However, the
participants in Chapuy's study were restricted to a specific
medical institution, which led to its classification as a lowquality RCT in our risk of bias assessment [25]. As explained in the sensitivity analysis section, after excluding
this low-quality RCT, the significant association between
combined oral vitamin D3 supplementation and calcium
and the reduction of PFF incidence in the geriatric population was no longer statistically significant. After excluding
low-quality studies, our findings are consistent with those
of the US Preventive Services Task Force (USPSTF). The
USPSTF concluded that current evidence is insufficient
to assess the balance of benefits and harms of combined
oral supplementation with doses greater than 400 IU of
vitamin D3 and greater than 1000 mg of calcium daily for
the primary prevention of osteoporotic fractures in community-dwelling, asymptomatic men and premenopausal
women [14].
Admittedly, our study also has some limitations. First,
the participants in the selected RCTs were predominantly
from developed countries in Europe and North America,
with a lack of relevant data from Asia and Africa, which
may affect the generalizability of the conclusions. Second,
some recent studies have suggested an association between vitamin D3 deficiency and the risk of PFF [33, 34];
however, we did not specifically include this subgroup of
individuals. Furthermore, it is important to consider factors that influence vitamin D3 absorption, especially in
the elderly population, as this could significantly affect the
efficacy of supplementation. In designing future studies,
it is important to consider a broader range of confounding
factors, such as the importance of monitoring changes in
blood levels of vitamin D in PFF patients. Finally, despite
the lack of evidence that vitamin D3 can reduce the incidence of PFF, vitamin D3 may still play a role in reducing
mortality from PFF and alleviating symptoms in PFF patients, which is a new direction for future research.
Conclusions
Oral supplementation with vitamin D3 alone in the elderly did not show a significant association with PFF incidence (OR = 1.04, 95% CI: 0.94-1.15, p-value = 0.46 > 0.05). Limited evidence suggested that combined oral supplementation with vitamin D3 and calcium may reduce the incidence of PFF in the elderly population (OR = 0.80, 95% CI: 0.69-0.94, p-value = 0.005 < 0.05), because after excluding low-quality RCTs, the protective effect was no longer statistically significant (OR = 0.90, 95% CI: 0.74- 1.10,p-value = 0.32 > 0.05). Therefore, there is a lack of robust evidence that oral vitamin D3 supplementation could reduce the incidence of PFF in the elderly population.
Declarations
Availability of data and materials
Not applicable.
Financial support and sponsorship
None.
Conflict of interest
All authors declared that there is no conflict of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
References
1. Sambrook P, & Cooper C. Osteoporosis. Lancet, 2006, 367(9527): 2010-2018. [Crossref]
2. Watts NB, & Manson JE. Osteoporosis and fracture risk evaluation and management: shared decision making in clinical practice. Jama, 2017, 317(3): 253-254. [Crossref]
3. Becker N, Hafner T, Pishnamaz M, Hildebrand F, & Kobbe P. Patient-specific risk factors for adverse outcomes following geriatric proximal femur fractures. Eur J Trauma Emerg Surg, 2022, 48(2): 753-761. [Crossref]
4. Klop C, Welsing PM, Cooper C, Harvey NC, Elders PJ, Bijlsma JW, et al. Mortality in British hip fracture patients, 2000-2010: a population-based retrospective cohort study. Bone, 2014, 66: 171-177. [Crossref]
5. Schnell S, Friedman SM, Mendelson DA, Bingham KW, & Kates SL. The 1-year mortality of patients treated in a hip fracture program for elders. Geriatr Orthop Surg Rehabil, 2010, 1(1): 6-14. [Crossref]
6. Bouillon R, Marcocci C, Carmeliet G, Bikle D, White JH, Dawson-Hughes B, et al. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev, 2019, 40(4): 1109-1151. [Crossref]
7. DeLuca HF. Overview of general physiologic features and functions of vitamin D. Am J Clin Nutr, 2004, 80(6 Suppl): 1689s-1696s. [Crossref]
8. Fahrleitner A, Dobnig H, Obernosterer A, Pilger E, Leb G, Weber K, et al. Vitamin D deficiency and secondary hyperparathyroidism are common complications in patients with peripheral arterial disease. J Gen Intern Med, 2002, 17(9): 663-669. [Crossref]
9. Thacher TD, & Clarke BL. Vitamin D insufficiency. Mayo Clin Proc, 2011, 86(1): 50-60. [Crossref]
10. Eleni A, & Panagiotis P. A systematic review and metaanalysis of vitamin D and calcium in preventing osteoporotic fractures. Clin Rheumatol, 2020, 39(12): 3571- 3579. [Crossref]
11. Hu ZC, Tang Q, Sang CM, Tang L, Li X, Zheng G, et al. Comparison of fracture risk using different supplemental doses of vitamin D, calcium or their combination: a network meta-analysis of randomised controlled trials. BMJ Open, 2019, 9(10): e024595. [Crossref]
12. Waterhouse M, Ebeling PR, McLeod DSA, English D, Romero BD, Baxter C, et al. The effect of monthly vitamin D supplementation on fractures: a tertiary outcome from the population-based, double-blind, randomised, placebo-controlled D-Health trial. Lancet Diabetes Endocrinol, 2023, 11(5): 324-332. [Crossref]
13. Uusi-Rasi K, Patil R, Karinkanta S, Kannus P, Tokola K, Lamberg-Allardt C, et al. Exercise and vitamin D in fall prevention among older women: a randomized clinical trial. JAMA Intern Med, 2015, 175(5): 703-711. [Crossref]
14. Grossman DC, Curry SJ, Owens DK, Barry MJ, Caughey AB, Davidson KW, et al. Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: US preventive services task force recommendation statement. Jama, 2018, 319(15): 1592-1599. [Crossref]
15. McGuinness LA, & Higgins JPT. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods, 2021, 12(1): 55-61. [Crossref]
16. Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. Bmj, 2019, 366: l4898. [Crossref]
17. Schwarzer G. meta: An R package for meta-analysis. R news, 2007, 7(3): 40-45.
18. Lips P, Graafmans WC, Ooms ME, Bezemer PD, & Bouter LM. Vitamin D supplementation and fracture incidence in elderly persons. A randomized, placebo-controlled clinical trial. Ann Intern Med, 1996, 124(4): 400-406. [Crossref]
19. Meyer HE, Smedshaug GB, Kvaavik E, Falch JA, Tverdal A, & Pedersen JI. Can vitamin D supplementation reduce the risk of fracture in the elderly? A randomized controlled trial. J Bone Miner Res, 2002, 17(4): 709-715. [Crossref]
20. Trivedi DP, Doll R, & Khaw KT. Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. Bmj, 2003, 326(7387): 469-479. [Crossref]
21. Grant AM, Avenell A, Campbell MK, McDonald AM, MacLennan GS, McPherson GC, et al. Oral vitamin D3 and calcium for secondary prevention of low-trauma fractures in elderly people (randomised evaluation of calcium or vitamin D, RECORD): a randomised placebocontrolled trial. Lancet, 2005, 365(9471): 1621-1628. [Crossref]
22. Lyons RA, Johansen A, Brophy S, Newcombe RG, Phillips CJ, Lervy B, et al. Preventing fractures among older people living in institutional care: a pragmatic randomised double blind placebo controlled trial of vitamin D supplementation. Osteoporos Int, 2007, 18(6): 811-818. [Crossref]
23. Smith H, Anderson F, Raphael H, Maslin P, Crozier S, & Cooper C. Effect of annual intramuscular vitamin D on fracture risk in elderly men and women--a populationbased, randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford), 2007, 46(12): 1852-1857. [Crossref]
24. Sanders KM, Stuart AL, Williamson EJ, Simpson JA, Kotowicz MA, Young D, et al. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. Jama, 2010, 303(18): 1815-1822. [Crossref]
25. Chapuy MC, Arlot ME, Delmas PD, & Meunier PJ. Effect of calcium and cholecalciferol treatment for three years on hip fractures in elderly women. Bmj, 1994, 308(6936): 1081-1082. [Crossref]
26. Chapuy MC, Pamphile R, Paris E, Kempf C, Schlichting M, Arnaud S, et al. Combined calcium and vitamin D3 supplementation in elderly women: confirmation of reversal of secondary hyperparathyroidism and hip fracture risk: the Decalyos II study. Osteoporos Int, 2002, 13(3): 257- 264. [Crossref]
27. Porthouse J, Cockayne S, King C, Saxon L, Steele E, Aspray T, et al. Randomised controlled trial of calcium and supplementation with cholecalciferol (vitamin D3) for prevention of fractures in primary care. Bmj, 2005, 330(7498): 1003-1013. [Crossref]
28. Jackson RD, LaCroix AZ, Gass M, Wallace RB, Robbins J, Lewis CE, et al. Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med, 2006, 354(7): 669-683. [Crossref]
29. Salovaara K, Tuppurainen M, Kärkkäinen M, Rikkonen T, Sandini L, Sirola J, et al. Effect of vitamin D(3) and calcium on fracture risk in 65- to 71-year-old women: a population-based 3-year randomized, controlled trial——the OSTPRE-FPS. J Bone Miner Res, 2010, 25(7): 1487-1495. [Crossref]
30. Bischoff-Ferrari HA, Willett WC, Orav EJ, Lips P, Meunier PJ, Lyons RA, et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N Engl J Med, 2012, 367(1): 40-49. [Crossref]
31. Avenell A, Mak JC, & O'Connell D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst Rev, 2014, 2014(4): Cd000227. [Crossref]
32. Manoj P, Derwin R, & George S. What is the impact of daily oral supplementation of vitamin D3 (cholecalciferol) plus calcium on the incidence of hip fracture in older people? A systematic review and meta-analysis. Int J Older People Nurs, 2023, 18(1): e12492. [Crossref]
33. Han J, Cho Y, Jee S, & Jo S. Vitamin D levels in patients with low-energy hip fractures. Hip Pelvis, 2020, 32(4): 192-198. [Crossref]
34. Kim HS, Jang G, Park JW, Lee YK, & Koo KH. Vitamin D deficiency and sarcopenia in hip fracture patients. J Bone Metab, 2021, 28(1): 79-83. [Crossref]